What is the name of the compound [Fe(H2O)5(SCN)]Cl2?
(a) pentaaquathiocyanatoiron(III) chloride
(b) pentaaquachlorothiocyanato iron(III)
(c) pentaaquathiocyanatoiron(III) dichloride
(d) pentaaquathiocyanatoiron(II) chloride

 Verified step by step guidance
Verified step by step guidance
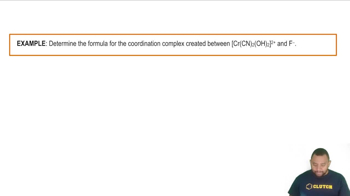
What is the name of the compound [Fe(H2O)5(SCN)]Cl2?
(a) pentaaquathiocyanatoiron(III) chloride
(b) pentaaquachlorothiocyanato iron(III)
(c) pentaaquathiocyanatoiron(III) dichloride
(d) pentaaquathiocyanatoiron(II) chloride
Based on effective nuclear charge (Zeff), which ion is the strongest oxidizing agent?
(a) Cu2+
(b) Ni2+
(c) Fe2+
(d) Mn2+
The [Cr(H2O)6]3+ ion is violet, and [Cr(CN)6]3- is yellow. Explain this difference using crystal field theory. Use the colors to order H2O and CN- in the spectrochemical series.
What is the oxidation state of the metal in each of the complexes?
a. [Ni(CN)5]3–
b. Ni(CO)4
c. [Co(en)2(H2O)Br]2+
d. [Cu(H2O)2(C2O4)2]2–
e. Co(NH3)3(NO2)3
Draw all possible diastereoisomers of [Cr(C2O4)2(H2O)2]-. Which can exist as a pair of enantiomers?
Predict the number of unpaired electrons for each of the following.
(c) Zn2+
(d) Cr3+