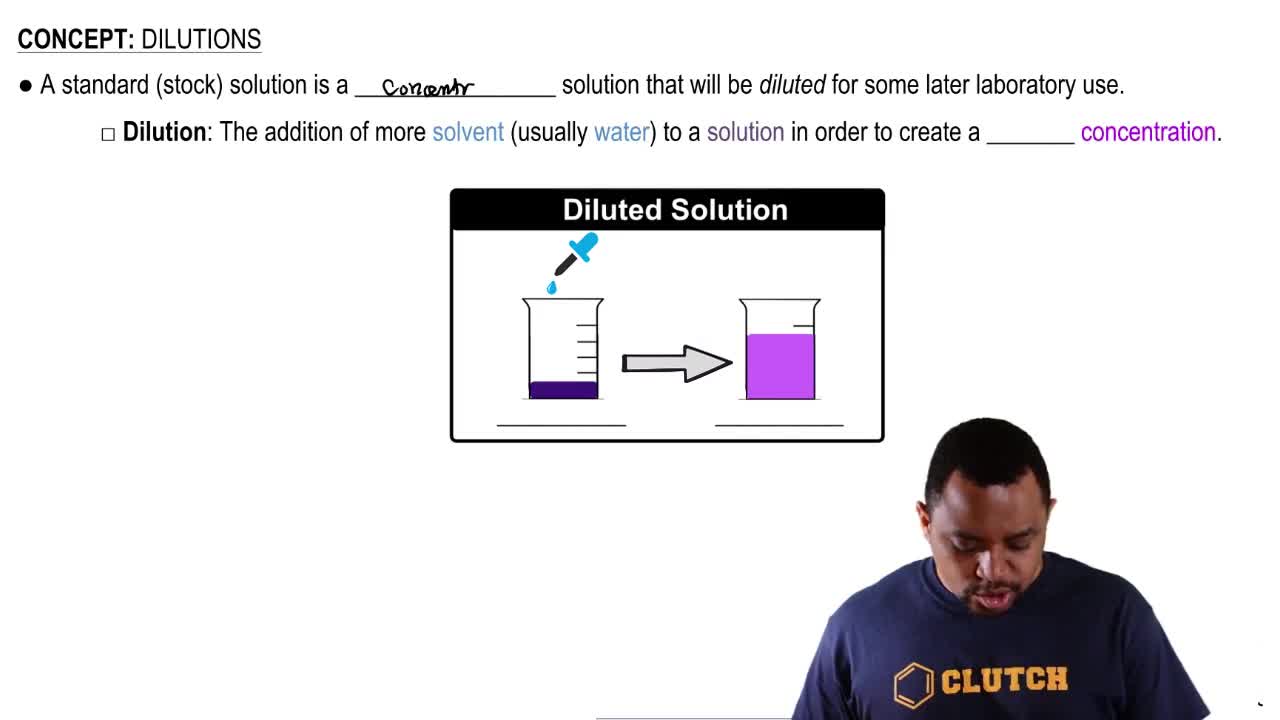
Molarity
Molarity (M) is a measure of concentration defined as the number of moles of solute per liter of solution. To prepare a solution, you need to know the desired molarity and the volume of the solution. In this case, a 0.325 M solution means there are 0.325 moles of benzoic acid in every liter of the solution. For 250 mL, you would calculate the moles needed based on this concentration.

 Verified step by step guidance
Verified step by step guidance