For each reaction, calculate ΔH°rxn, ΔS°rxn, and ΔG°rxn at 25 °C and state whether or not the reaction is spontaneous. If the reaction is not spontaneous, would a change in temperature make it spontaneous? If so, should the temperature be raised or lowered from 25 °C? a. N2O4(g) → 2 NO2(g)

For each reaction, calculate ΔH°_rxn, ΔS°_rxn, and ΔG°_rxn at 25°C and determine whether the reaction is spontaneous. If the reaction is not spontaneous, could a change in temperature make it spontaneous? If so, should the temperature be increased or decreased from 25°C? b. 2 NH3(g) → N2H4(g) + H2(g)
 Verified step by step guidance
Verified step by step guidanceKey Concepts
Enthalpy (ΔH°)

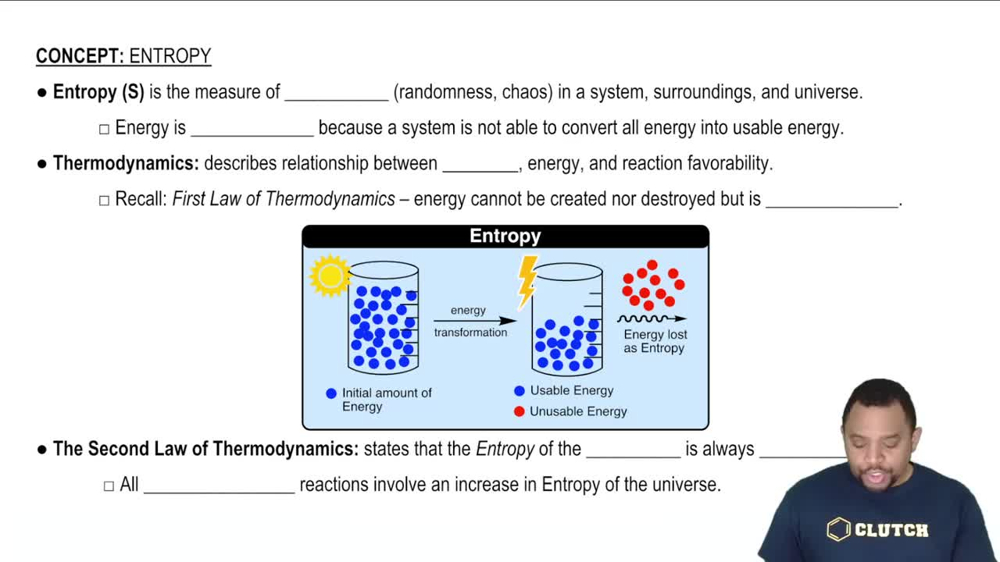
Entropy (ΔS°)
Gibbs Free Energy (ΔG°)

For each reaction, calculate ΔH°rxn, ΔS°rxn, and ΔG°rxn at 25 °C and state whether or not the reaction is spontaneous. If the reaction is not spontaneous, would a change in temperature make it spontaneous? If so, should the temperature be raised or lowered from 25 °C? d. N2(g) + 3 H2(g) → 2 NH3(g)
For each reaction, calculate ΔH°rxn, ΔS°rxn, and ΔG°rxn at 25 °C and state whether or not the reaction is spontaneous. If the reaction is not spontaneous, would a change in temperature make it spontaneous? If so, should the temperature be raised or lowered from 25 °C? a. 2 CH4(g) → C2H6(g) + H2(g)
For each reaction, calculate ΔH°rxn, ΔS°rxn, and ΔG°rxn at 25 °C and state whether or not the reaction is spontaneous. If the reaction is not spontaneous, would a change in temperature make it spontaneous? If so, should the temperature be raised or lowered from 25 °C? c. N2(g) + O2(g) → 2 NO(g)
For each reaction, calculate ΔH°rxn, ΔS°rxn, and ΔG°rxn at 25 °C and state whether or not the reaction is spontaneous. If the reaction is not spontaneous, would a change in temperature make it spontaneous? If so, should the temperature be raised or lowered from 25 °C? d. 2 KClO3(s) → 2 KCl(s) + 3 O2(g)
