 Back
BackWeek 5 class slides
Study Guide - Smart Notes

Tissue Culture and Plant Transformation Systems
Introduction to Tissue Culture
Tissue culture is a set of techniques used to grow plant cells, tissues, or organs under sterile conditions on a nutrient culture medium. It plays a crucial role in plant biotechnology, allowing for the propagation of rare or endangered plants and the production of valuable compounds such as vaccines, antibodies, or enzymes in plant cells or tissues.
Definition: The in vitro cultivation of plant cells, tissues, or organs on nutrient media under controlled conditions.
Applications: Agriculture (mass propagation, conservation), medicine (production of pharmaceuticals), and industry (enzyme production).
Example: Propagation of disease-free banana plants using tissue culture.

Overview and Methods of Tissue Culture
Tissue culture involves several steps, from the selection of explants to the acclimatization of plantlets. The process can be divided into organogenesis (formation of organs) and callogenesis (formation of callus tissue), each with specific applications.
Organogenesis: Leads to micropropagation and hairy root culture.
Callogenesis: Involves cell suspension cultures and genetic transformation.

Process of Tissue Culture
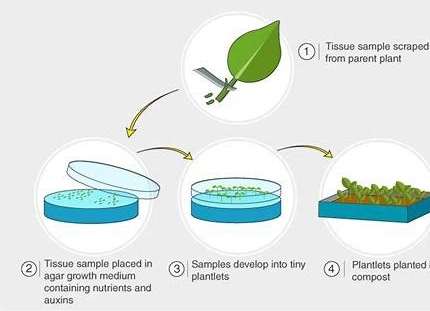
The tissue culture process consists of several key steps:
Explant selection: Choosing healthy, young, disease-free plant material.
Sterilization: Removing microorganisms using ethanol or bleach.
Culture media preparation: Preparing nutrient-rich media with hormones like auxins, cytokinins, and gibberellins.
Explant inoculation: Placing sterilized explants onto the growth media, leading to callus formation.
Incubation and growth: Allowing explants to grow under controlled conditions.
Subculturing: Transferring calli to new media to induce shoot growth.
Rooting: Transferring shoots to rooting medium.
Acclimatization: Gradually exposing plantlets to external conditions before potting.


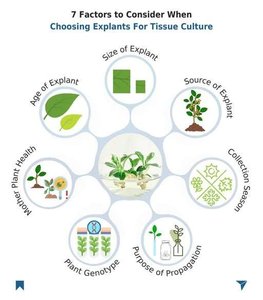
Factors Affecting Explant Selection
Several factors influence the success of tissue culture, including the age, size, and source of the explant, as well as the health and genotype of the mother plant.
Mother plant health
Age and size of explant
Purpose of propagation
Collection season
Advantages and Disadvantages of Tissue Culture
Tissue culture offers several benefits but also presents challenges.
Advantages:
Mass propagation of plants with desired qualities
Conservation of endangered species
Production of disease-free plants
Disadvantages:
High costs
Genetic instability
Technical expertise required
Plant Genetic Transformation Systems
Overview of Plant Transformation
Plant transformation systems introduce foreign DNA into plant cells or tissues, creating genetically modified plants with traits such as pest resistance or environmental stress tolerance.
Purpose: To enhance crop traits, improve yield, and confer resistance to diseases or environmental stresses.

Modes of Plant Transformation
There are several methods for introducing foreign DNA into plant cells:
Agrobacterium tumefaciens-mediated transformation: Utilizes the natural ability of Agrobacterium to transfer DNA to plant cells.
Electroporation: Uses electrical pulses to introduce DNA into plant protoplasts.
Gene gun (particle bombardment): Physically delivers DNA-coated particles into plant cells.
Microfibers: Uses fine fibers to introduce DNA into cells.
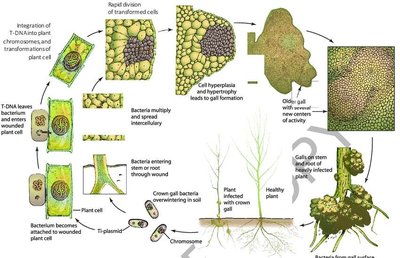
Agrobacterium tumefaciens-Mediated Transformation
This method exploits the bacterium's Ti plasmid, which naturally integrates into the plant genome, causing crown gall disease. Scientists modify the Ti plasmid to carry desired genes instead of disease genes.

Electroporation, Gene Gun, and Microfibers
Alternative methods are used for plants less susceptible to Agrobacterium infection or for direct DNA delivery.
Electroporation: Protoplasts are exposed to an electric field, creating temporary pores for DNA uptake.
Gene gun: DNA-coated gold or tungsten particles are shot into plant tissues.
Microfibers: Physical penetration of cell walls with DNA-coated fibers.
Challenges in Plant Transformation
Efficient delivery and integration of the new gene into the plant genome
Survival and regeneration of transformed cells
Identification and selection of transformed cells
Ensuring the transgene does not disrupt essential genes or regulatory elements
CRISPR/Cas9: Gene Editing Technology
Origin and Mechanism of CRISPR/Cas9
CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats) is a bacterial immune system adapted for genome editing. The system uses a guide RNA to direct the Cas9 nuclease to a specific DNA sequence, where it introduces a double-stranded break.
Components: crRNA, tracrRNA (or single guide RNA, sgRNA), Cas9 protein, and a PAM sequence.
Function: Targeted DNA cleavage and subsequent repair by the cell's machinery.
Modernization of CRISPR-Cas
In 2012, Jennifer Doudna and Emmanuelle Charpentier developed the CRISPR-Cas9 system for genome editing in eukaryotes, earning the Nobel Prize in Chemistry in 2020. The system was simplified by linking crRNA and tracrRNA into a single guide RNA (sgRNA).
Double-stranded break repair mechanisms:
Non-homologous end joining (NHEJ): Error-prone, leads to insertions or deletions (indels).
Homology-directed repair (HDR): Uses a donor template for precise editing.
Limitations and Applications of CRISPR/Cas9
Limitations: Off-target effects, delivery challenges, immune responses, and efficiency issues.
Applications: Gene knockout, gene correction, disease modeling, and crop improvement.
DNA Sequencing and Genomic Analysis
Introduction to Genomics, Transcriptomics, and Proteomics
These fields study the complete sets of DNA, RNA, and proteins in an organism, respectively, providing insights into genetic variation, gene expression, and cellular function.
Genomics: Study of the entire genome.
Transcriptomics: Study of all RNA transcripts.
Proteomics: Study of all proteins expressed.
Metabolomics: Study of all metabolites present.
DNA Sequencing Techniques
DNA sequencing determines the order of nucleotide bases in DNA. There are three main generations of sequencing technologies:
First-generation (Sanger sequencing): Uses dideoxynucleotides to terminate DNA synthesis at specific bases, generating fragments of varying lengths.
Second-generation (Next-Generation Sequencing, NGS): Massively parallel sequencing, e.g., Illumina sequencing, allows for high-throughput and cost-effective sequencing.
Third-generation (Single-Molecule Real-Time, SMRT): Sequences single DNA molecules in real time, e.g., PacBio and Nanopore sequencing, enabling longer reads.
Comparison of Sequencing Technologies
Technology | Read Length | Throughput | Accuracy | Applications |
|---|---|---|---|---|
Sanger | Up to 700 bp | Low | High | Small-scale, validation |
Illumina (NGS) | 100-300 bp | High | High | Whole-genome, transcriptome |
PacBio/Nanopore | 10,000+ bp | Moderate | Moderate | Structural variation, long reads |
Genome Mapping
Genome mapping identifies the locations of genes and markers on chromosomes. Genetic maps are based on recombination rates, while physical maps use direct DNA analysis to measure distances in base pairs.
Genetic maps: Constructed from linkage analysis, provide relative gene positions.
Physical maps: Measure actual distances in base pairs, kilobases, or megabases.
Single Nucleotide Polymorphisms (SNPs) and Genome-Wide Association Studies (GWAS)
SNPs are single base pair variations in the genome. They serve as genetic markers for studying inheritance and disease associations.
Haplotype: A set of SNPs and other variants inherited together.
Tag-SNP: Representative SNPs used to identify haplotypes.
Linkage disequilibrium: Nonrandom association of alleles at different loci.
GWAS: Studies that use SNPs to find genetic variants associated with diseases.
Applications of Metagenomics
Metagenomics involves sequencing the collective genomes of microbial communities from environmental samples, enabling the study of unculturable microbes and community structure.
Applications: Food safety, environmental monitoring, medical diagnostics, and industrial biotechnology.
