 Back
BackMembrane-Based Energy Generation and Mitochondrial Function: Study Notes for Genetics Students
Study Guide - Smart Notes

Membrane-Based Mechanisms of Energy Generation
Overview of ATP Production
ATP is the primary energy currency in cells, and most of it is generated through membrane-based processes such as oxidative phosphorylation and photosynthesis. These processes rely on electron transport chains embedded in cellular membranes to create a proton gradient, which is then used to synthesize ATP.
Oxidative phosphorylation: Occurs in mitochondria of eukaryotes, using energy from food.
Photosynthesis: Occurs in chloroplasts of plants and green algae, harnessing solar energy.
Electron transport chain (ETC): Series of protein complexes that transfer electrons and pump protons across membranes.
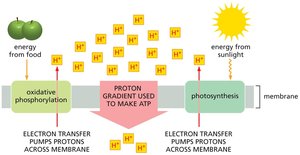
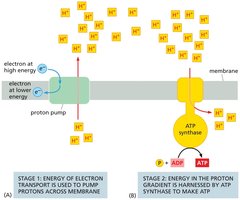
Chemiosmotic Coupling
Chemiosmotic coupling links the energy from electron transport to ATP synthesis. It involves two stages: electron transport and ATP synthesis via ATP synthase.
Stage 1: High-energy electrons are transferred along the ETC, pumping protons across the membrane.
Stage 2: Protons flow back through ATP synthase, driving the conversion of ADP and Pi to ATP.
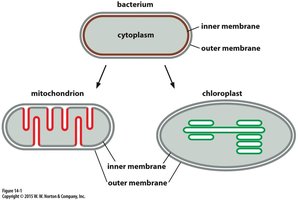
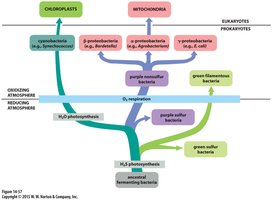
Evolutionary Origins of Mitochondria and Chloroplasts
Shared Features with Bacteria
Mitochondria and chloroplasts are believed to have evolved from ancestral bacteria through endosymbiosis. They retain several bacterial features:
Reproduction by fission
Circular genomes
Bacterial-like RNA and protein synthesis machinery
Many genes transferred to the nuclear genome
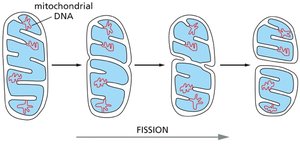
Mitochondrial Structure and Dynamics
Dynamic Nature of Mitochondria
Mitochondria are highly dynamic, adjusting their location, shape, and number according to cellular energy demands. They can remain fixed or form networks, and undergo fission and fusion.
Fixed location: E.g., near contractile apparatus in heart muscle cells.
Network formation: Tubular structures distributed throughout cytoplasm.
Fission and fusion: Mitochondria can break apart and reform.
Variable numbers: From few to thousands per cell.
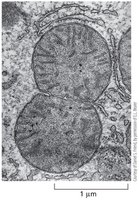
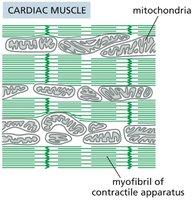
Specialized Mitochondrial Localization
Mitochondria are strategically positioned in cells to meet energy requirements.
Cardiac muscle: Mitochondria are located near myofibrils for efficient ATP supply.
Sperm: Mitochondria wrap around the flagellum to power motility.
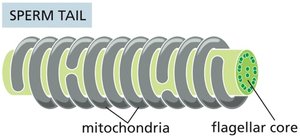
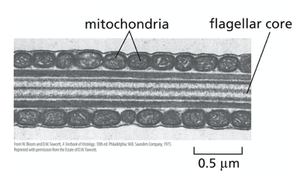

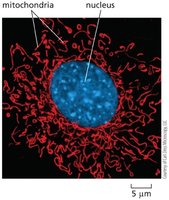
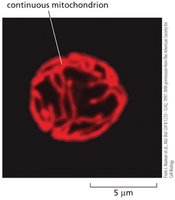
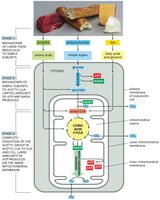
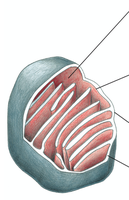
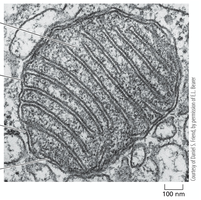
Mitochondrial Compartments
Mitochondria are organized into four distinct compartments, each with specialized functions.
Outer membrane: Contains porins, permeable to molecules <5000 daltons.
Inner membrane: Impermeable to ions, highly folded (cristae), contains ETC and ATP synthase.
Intermembrane space: Space between inner and outer membranes.
Matrix: Contains enzymes for citric acid cycle, genome replication, and protein synthesis.
Citric Acid Cycle and Electron Transport Chain
Citric Acid Cycle (Krebs Cycle)
The citric acid cycle is a central metabolic pathway in the mitochondrial matrix, generating high-energy electrons for ATP production.
Inputs: Pyruvate and fatty acids (from glycolysis and fat breakdown).
Process: Acetyl-CoA is oxidized to CO2, producing NADH and FADH2.
Outputs: High-energy electrons carried by NADH and FADH2 to the ETC.
Electron Transport Chain (ETC)
NADH and FADH2 donate electrons to the ETC, which consists of three main complexes in the inner mitochondrial membrane:
NADH dehydrogenase complex
Cytochrome c reductase complex
Cytochrome c oxidase complex
Electrons are transferred stepwise, releasing energy to pump protons across the membrane, creating an electrochemical gradient.
ATP Synthesis and Chemiosmotic Mechanism
Proton Gradient and ATP Synthase
The electrochemical proton gradient across the inner mitochondrial membrane is used by ATP synthase to produce ATP.
ATP synthase: Large protein complex embedded in the inner membrane.
Mechanism: Protons flow through ATP synthase, driving the conversion of ADP and Pi to ATP.
Efficiency: Each NADH yields ~2.5 ATP; each FADH2 yields ~1.5 ATP.
Reversibility and Transport
ATP synthase can operate in reverse, using ATP hydrolysis to pump protons. The proton gradient also drives the transport of molecules across the inner membrane.
Experimental Evidence and Redox Chemistry
Chemiosmotic Hypothesis
Experimental evidence supports the chemiosmotic hypothesis, showing that the electrochemical proton gradient is essential for ATP synthesis.
Redox Potential and Electron Carriers
Electron transport depends on redox reactions and the relative electron affinities of carriers. Key carriers include flavins, iron-sulfur clusters, ubiquinone, cytochromes, and copper centers.
Summary Table: Mitochondrial Compartments and Functions
Compartment | Main Features | Function | |
|---|---|---|---|
Outer membrane | Porins, permeable to small molecules | Allows passage of metabolites | |
Inner membrane | Impermeable, cristae, ETC, ATP synthase | Site of oxidative phosphorylation | |
Intermembrane space | Between membranes | H+ accumulation for gradient | |
Matrix | Enzymes, genome, ribosomes | Citric acid cycle, protein synthesis |
Key Equations
ATP synthesis:
Citric acid cycle electron carriers:
Reduction of oxygen:
Additional info:
Mitochondrial and chloroplast genomes are circular, similar to bacterial genomes.
Transition metal ions and quinones are essential for electron transport due to their variable oxidation states.
Proton gradients are fundamental to both cellular respiration and photosynthesis.
