 Back
BackMutation and DNA Repair: Mechanisms, Causes, and Consequences
Study Guide - Smart Notes

Mutation: Causes and Types
Spontaneous vs. Induced Mutations
Mutations are changes in the nucleotide sequence of DNA and can arise spontaneously or be induced by external factors. Understanding their origin is crucial for genetics, as mutations are the source of genetic variation and can lead to disease.
Spontaneous mutations: Occur naturally due to normal biological or chemical processes, such as errors during DNA replication or base damage.
Induced mutations: Result from exposure to external agents (mutagens) like radiation, chemicals, or UV light.
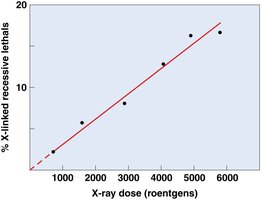
Mutation rates: Spontaneous mutation rates are generally low, but vary among organisms and loci.
Organism | Character | Locus | Rate |
|---|---|---|---|
Bacteriophage T2 | Lysis inhibition | r+ → r | 1 × 10-8 |
Escherichia coli | Lactose fermentation | lac+ → lac- | 4 × 10-9 |
Zea mays | Shrunken seeds | sh+ → sh | 1 × 10-6 |
Drosophila melanogaster | White eye | w+ → w | 1 × 10-7 |
Mus musculus | Brown coat | B+ → b | 3.5 × 10-7 |

Spontaneous Mutations: Mechanisms
Replication Errors and Slippage
DNA replication is not perfect; errors can occur when DNA polymerase inserts incorrect nucleotides or when slippage happens in repetitive sequences.
Point mutations: Result from mispairing during replication.
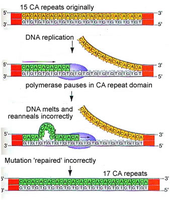
Replication slippage: Occurs when DNA polymerase misses looped-out nucleotides, leading to insertions or deletions, especially in repeat sequences (mutation hot spots).
Hereditary diseases: Replication slippage contributes to disorders like Fragile-X and Huntington disease.
Tautomeric Shifts
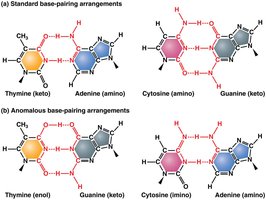
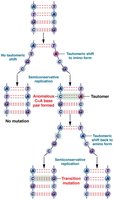
Purines and pyrimidines can exist in alternate chemical forms called tautomers, which increase the likelihood of mispairing during DNA replication.
Tautomeric shifts: Change the bonding structure, allowing noncomplementary base pairing and potentially leading to permanent mutations.
Example: Thymine (keto) can shift to thymine (enol), pairing incorrectly with guanine.
DNA Base Damage: Depurination and Deamination
Spontaneous mutations can also arise from chemical changes to DNA bases, such as depurination and deamination.
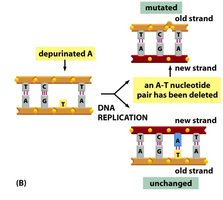
Depurination: Loss of a purine base (adenine or guanine), creating an apurinic site. During replication, the missing base is often skipped, resulting in a deletion.
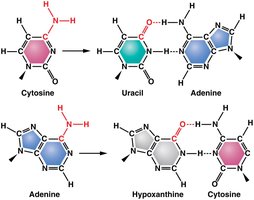
Deamination: Removal of an amino group from cytosine or adenine, converting cytosine to uracil and adenine to hypoxanthine. This alters base pairing, e.g., A=T converted to G=C.
Oxidative Damage and Free Radicals
Oxidative damage to DNA is caused by reactive oxygen species (ROS) generated during normal cellular metabolism or exposure to radiation.
Free radicals: Molecules with unpaired electrons that can damage DNA by altering bases, breaking phosphodiester bonds, and causing deletions or translocations.
Prevention: Antioxidants in the diet can reduce free radical levels.
Induced Mutations: Chemical and Physical Agents
Mutagens
Mutagens are agents that increase the frequency of mutations. They include chemicals, radiation, and biological factors.
Examples: Fungal toxins, cosmic rays, UV light, industrial pollutants, X-rays, chemicals in tobacco smoke.
Base Analogs
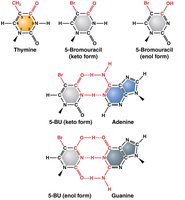
Base analogs are chemicals that resemble DNA bases and can be incorporated during DNA synthesis, increasing mutation rates.
Example: 5-Bromouracil acts as a thymine analog, increasing tautomeric shifts and sensitivity to UV light.
Alkylating Agents
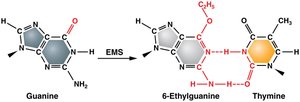
Alkylating agents donate alkyl groups to DNA bases, altering their pairing properties and causing transition mutations.
Example: Mustard gas and ethylmethanosulfonate (EMS) are commonly used in genetic experiments.
Intercalating Agents
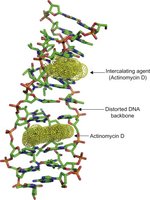
Intercalating agents are chemicals that insert themselves between DNA base pairs, causing distortions and replication errors.
Example: Ethidium bromide and acridine dyes.
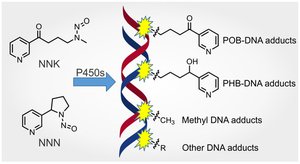
Adduct-Forming Agents
Adduct-forming agents covalently bind to DNA, altering its conformation and interfering with replication and repair.
Examples: Acetaldehyde (cigarette smoke), heterocyclic amines (cooked meats).
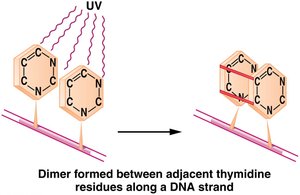
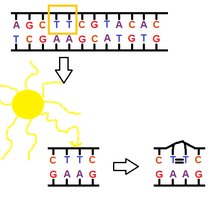
UV Light and Pyrimidine Dimers
UV radiation induces mutations by creating pyrimidine dimers, which distort DNA and interfere with replication.
Pyrimidine dimers: Two adjacent pyrimidines (usually thymine) become covalently linked, causing replication errors.
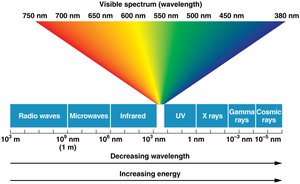
Ionizing Radiation
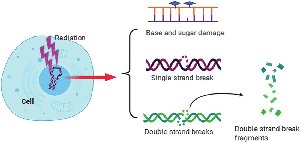
Ionizing radiation (X-rays, gamma rays, cosmic rays) penetrates tissues and causes ionization of molecules, leading to DNA damage.
Effects: Breaks in DNA strands, base damage, chromosomal rearrangements.
DNA Repair Mechanisms
Proofreading and Mismatch Repair
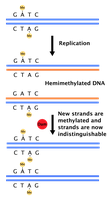
DNA polymerase proofreads newly synthesized DNA, removing and replacing incorrect nucleotides. If proofreading fails, mismatch repair systems are activated.
Strand discrimination: Based on DNA methylation; newly synthesized strands are unmethylated and recognized for repair.
Enzymes: Exonucleases and endonucleases remove mismatches; DNA polymerase inserts correct nucleotides.
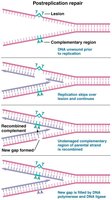
Postreplication Repair
Postreplication repair responds to DNA damage that escapes initial repair and replication. RecA protein directs recombination with undamaged parental DNA to fill gaps.
SOS Repair System in E. coli
The SOS repair system is a last-resort mechanism in bacteria, allowing replication to proceed despite DNA damage. It is error-prone and can itself be mutagenic.
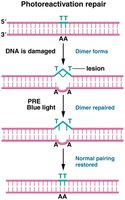
Photoreactivation Repair
Photoreactivation repair (PRE) uses an enzyme that absorbs light to cleave bonds between thymine dimers, reversing UV-induced damage. This mechanism is absent in humans.
Base and Nucleotide Excision Repair
Excision repair mechanisms remove damaged bases or nucleotides and replace them with correct ones. Two main types:
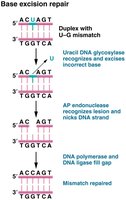
Base excision repair (BER): Corrects single damaged bases; DNA glycosylase recognizes and removes the altered base.
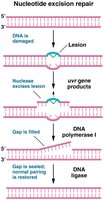
Nucleotide excision repair (NER): Repairs bulky lesions that distort the DNA helix; endonuclease cuts out the error, DNA polymerase fills the gap, and DNA ligase seals the nick.
Xeroderma Pigmentosum (XP)
XP is a rare genetic disorder caused by defects in NER pathways. Affected individuals have severe skin abnormalities, high cancer risk, and neurological defects.

Double-Strand Break Repair
Double-strand breaks are highly dangerous and can lead to chromosomal rearrangements, cancer, or cell death. Two main repair pathways:
Homologous recombination repair: Uses a sister chromatid as a template to accurately repair the break.
Nonhomologous end joining: Directly ligates the broken ends, often resulting in mutations.
Transposable Elements
Transposons and Their Effects
Transposable elements (TEs) are DNA sequences that can move within and between genomes, causing mutations and chromosomal rearrangements.
Types: Autonomous (can move by themselves) and nonautonomous (require help from autonomous elements).
Effects: Disrupt protein-coding regions, alter gene expression, and contribute to genetic diversity.
Transposable Elements in Drosophila and Humans
Many families of TEs exist in Drosophila (e.g., Copia, P elements) and humans (e.g., LINEs, SINEs). P elements are used as genetic tools in Drosophila, and TEs contribute to human genome variability.
Additional info: Transposons may contribute to evolution by generating genetic diversity and facilitating adaptation.
