 Back
BackCh 9 art 2 for exam 2
Study Guide - Smart Notes

Ch. 9 – The Molecular Biology of Translation
Introduction to Translation
Translation is the process by which the genetic information encoded in messenger RNA (mRNA) is used to assemble a specific sequence of amino acids, forming a polypeptide (protein). This process is fundamental to gene expression and is highly conserved across all domains of life.
The Relationship Between Genes and Proteins
Gene and Protein Sequence Correspondence
Genes are composed of DNA sequences that are transcribed into mRNA.
The coding strand of DNA has the same sequence as the mRNA (except T is replaced by U in RNA).
The template strand is used by RNA polymerase to synthesize the complementary mRNA.
Each group of three nucleotides (codon) in mRNA specifies one amino acid in the protein.
Example: For the DNA coding sequence ATG GGG CCC AAT GAA, the mRNA is AUG GGG CCC AAU GAA, which translates to the protein sequence: Met-Gly-Pro-Asn-Glu.
The Genetic Code
Features of the Genetic Code
The genetic code is universal with few exceptions, meaning it is used by almost all organisms.
It is composed of triplet codons, each specifying one amino acid.
The code is degenerate: multiple codons can code for the same amino acid.
There are start (AUG) and stop (UAA, UAG, UGA) codons that signal initiation and termination of translation.
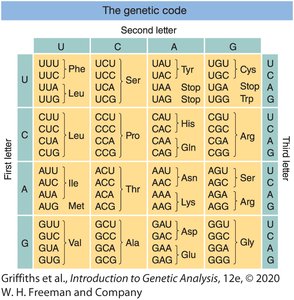
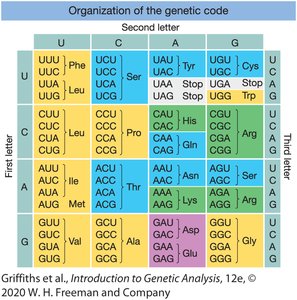
Organization of the Genetic Code
The code can be visualized as a table with the first, second, and third bases of the codon determining the amino acid.
Codons are read in the 5' to 3' direction on the mRNA.
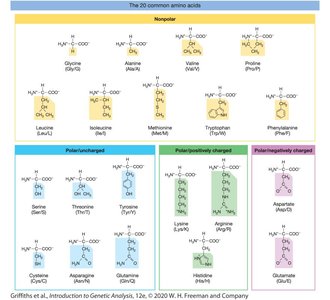
Chemical Properties of Amino Acids
Amino acids can be classified as nonpolar, polar uncharged, polar positively charged, or polar negatively charged.
The chemical properties of amino acids influence protein structure and function.
tRNA and Aminoacyl-tRNA Synthetases
tRNA Structure and Function
Transfer RNA (tRNA) molecules serve as adaptors that match amino acids to their corresponding codons in mRNA.
Each tRNA has an anticodon that base-pairs with a codon on the mRNA and an acceptor stem that attaches to a specific amino acid.
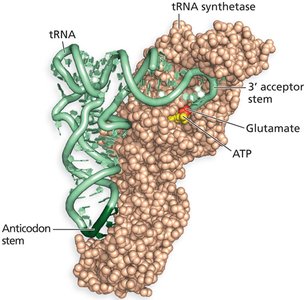
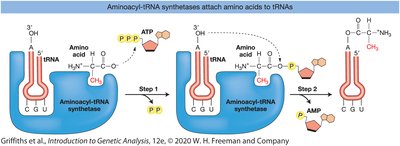
Aminoacyl-tRNA Synthetases
These enzymes attach the correct amino acid to its corresponding tRNA, a process called charging the tRNA.
Proofreading by these enzymes ensures high fidelity in protein synthesis.
Codon-Anticodon Interaction and the Wobble Hypothesis
Base Pairing Rules
Codon-anticodon pairing follows standard Watson-Crick base pairing (A-U, G-C) but is antiparallel (mRNA 5'→3', tRNA 3'→5').
The third position of the codon (the "wobble" position) allows for non-standard base pairing, increasing the efficiency of translation.
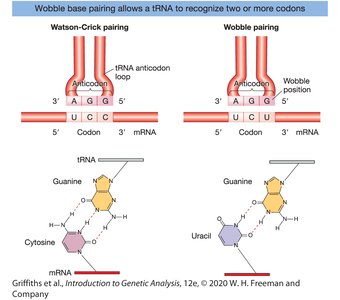
The Wobble Position
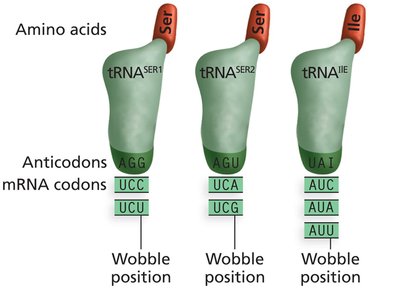
Inosine (I) in the tRNA anticodon can pair with U, C, or A in the mRNA codon.
This flexibility allows one tRNA to recognize multiple codons for the same amino acid.
Wobble Base-Pairing Rules Table
3′ Nucleotide of Codon | 5′ Nucleotide of Anticodon |
|---|---|
A or G | U |
G | C |
U | A |
U or C | G |
U, C, or A | I |
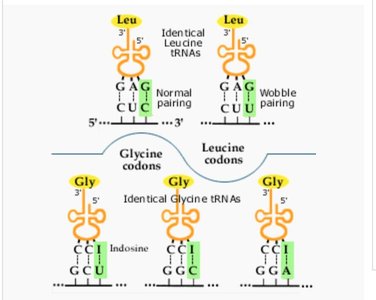
Examples of Wobble Pairing
Some tRNAs can pair with more than one codon due to wobble at the third position.
Post-Translational Modifications
Types and Importance
After translation, proteins often undergo post-translational modifications such as phosphorylation, methylation, and adenylation.
These modifications are critical for protein function, localization, and regulation.
Proteins may also be cleaved to become active or to remove targeting sequences.
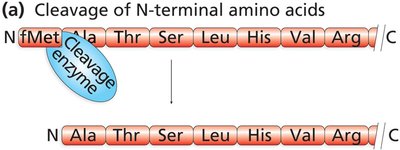
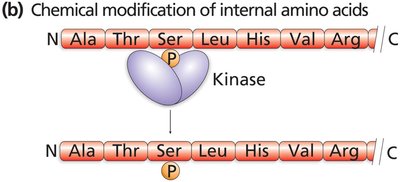
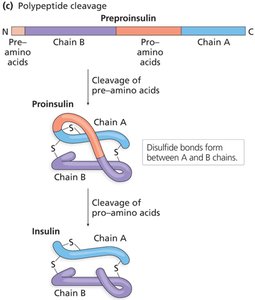
Translation in Eukaryotes: Protein Targeting and Processing
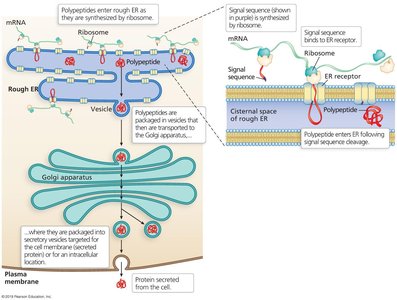
Targeting to the Rough Endoplasmic Reticulum (ER)
Proteins destined for secretion or for certain organelles are targeted to the rough ER by a signal sequence.
During translation, the ribosome-mRNA complex binds to the ER, and the growing polypeptide is translocated into the ER lumen.
Proteins may then be transported to the Golgi apparatus for further modification and sorting.
Summary Table: Key Concepts in Translation
Concept | Description |
|---|---|
Genetic Code | Triplet codons in mRNA specify amino acids |
tRNA | Adaptor molecule with anticodon and amino acid attachment site |
Aminoacyl-tRNA Synthetase | Enzyme that charges tRNA with correct amino acid |
Wobble Hypothesis | Flexibility in third codon position allows one tRNA to recognize multiple codons |
Post-Translational Modification | Chemical changes to proteins after synthesis, essential for function |
Protein Targeting | Signal sequences direct proteins to correct cellular locations |
Review and Concept Integration
Translation is tightly linked to transcription and DNA replication in the central dogma of molecular biology.
Understanding the genetic code and translation mechanisms is essential for genetic engineering, biotechnology, and medicine.
