 Back
BackAcids, Bases, and Equilibrium: Comprehensive Study Notes for Chapter 10
Study Guide - Smart Notes

Acids and Bases: Fundamental Concepts
Types and Definitions of Acids and Bases
Acids and bases are central to chemical reactions and biological processes. Their definitions vary depending on the theoretical framework:
Arrhenius Acid: Produces hydronium ions (H3O+) when dissolved in water.
Brønsted-Lowry Acid: Proton donor.
Lewis Acid: Electron pair acceptor.
Arrhenius Base: Produces hydroxide ions (OH-) in water.
Brønsted-Lowry Base: Proton acceptor.
Lewis Base: Electron pair donor.
Acids are typically sour, turn litmus red, and neutralize bases. Bases are bitter, slippery, turn litmus blue, and neutralize acids.
Acids and Bases in Water
Acids and bases dissociate in water to produce ions. For example:
HCl(g) → H+(aq) + Cl-(aq)
NaOH(s) → Na+(aq) + OH-(aq)
Lone protons (H+) do not exist freely in solution; they are always bound to water, forming hydronium ions (H3O+).
Naming Acids and Bases
Acids are named based on their composition:
Binary Acids: Prefix "hydro-" and suffix "-ic acid" (e.g., HCl: hydrochloric acid).
Oxoacids: Suffix changes from "-ate" to "-ic acid" or "-ite" to "-ous acid" (e.g., H2SO4: sulfuric acid).
Bases with OH- are named as the hydroxide of the metal (e.g., NaOH: sodium hydroxide).
Acid-Base Reactions and Conjugate Pairs
Brønsted-Lowry Theory and Amphoteric Nature of Water
Acids donate protons, bases accept protons. Water can act as both an acid and a base, making it amphoteric.
HCl + H2O → H3O+ + Cl-
NH3 + H2O → NH4+ + OH-
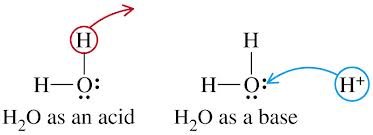
Conjugate Acid-Base Pairs
Every acid-base reaction involves two conjugate pairs, related by the loss and gain of H+:
HA + B ↔ A- + BH+
Example: HF + H2O ↔ F- + H3O+
Chemical Equilibrium and Le Chatelier’s Principle
Equilibrium Concepts
Chemical equilibrium is a dynamic state where reactant and product concentrations remain constant. Reversible reactions reach equilibrium when the rates of forward and reverse reactions are equal.
Example: N2 + 3H2 ↔ 2NH3
Le Chatelier’s Principle
If a stress (change in concentration, temperature, or pressure) is applied to a system at equilibrium, the system shifts to relieve that stress.
Adding reactants shifts equilibrium toward products.
Removing products shifts equilibrium toward products.
A catalyst speeds up attainment of equilibrium but does not affect the equilibrium position.
Acid and Base Strength
Strong vs. Weak Acids and Bases
Strong acids and bases dissociate completely in water, producing high concentrations of ions. Weak acids and bases dissociate only partially, resulting in lower ion concentrations.
Strong acid example: HCl
Weak acid example: CH3COOH (acetic acid)
Ionization of Water and pH
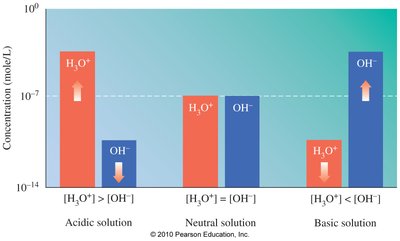
Ion Product of Water (Kw)
Water ionizes to produce equal concentrations of H3O+ and OH- in pure water:
[H3O+] = [OH-] = 1.0 × 10-7 M
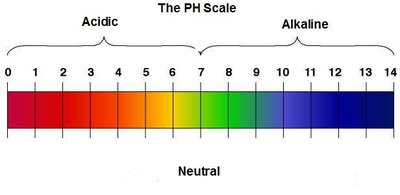
pH Scale and Calculations
The pH scale measures acidity and alkalinity, ranging from 0 (most acidic) to 14 (most basic). Neutral solutions have pH 7.
For [H3O+] = 1 × 10-4 M, pH = 4
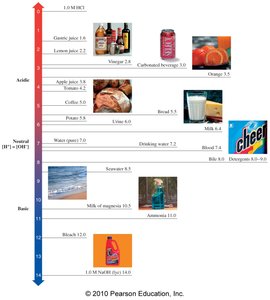
pH of Everyday Substances
Common substances have characteristic pH values, from acidic (lemon juice, vinegar) to basic (bleach, ammonia).
Comparison Table: [H3O+], [OH-], and pH
This table summarizes the relationship between hydronium ion concentration, hydroxide ion concentration, and pH at 25°C:
[H3O+] | pH | [OH-] |
|---|---|---|
100 | 0 | 10-14 |
10-1 | 1 | 10-13 |
10-2 | 2 | 10-12 |
10-3 | 3 | 10-11 |
10-4 | 4 | 10-10 |
10-5 | 5 | 10-9 |
10-6 | 6 | 10-8 |
10-7 | 7 | 10-7 |
10-8 | 8 | 10-6 |
10-9 | 9 | 10-5 |
10-10 | 10 | 10-4 |
10-11 | 11 | 10-3 |
10-12 | 12 | 10-2 |
10-13 | 13 | 10-1 |
10-14 | 14 | 100 |

Acid-Base Neutralization and Titration
Neutralization Reactions
Acids react with bases to produce a salt and water. For example:
HCl(aq) + NaOH(aq) → NaCl(aq) + H2O(l)
2HCl(aq) + Ca(OH)2(aq) → CaCl2(aq) + 2H2O(l)
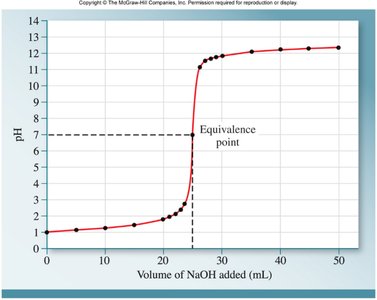
Titration and Calculating Molarity
Titration is used to determine the concentration of an acid or base by neutralization. The equivalence point is where the amount of titrant added is stoichiometrically equal to the substance being analyzed.
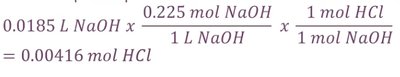

Buffer Solutions
Buffer Action and Components
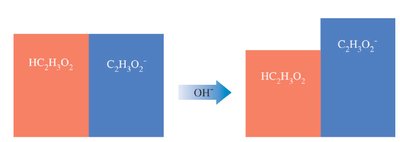
Buffers resist changes in pH when acids or bases are added. They consist of a weak acid and its conjugate base, or a weak base and its conjugate acid.
Example: Acetic acid (HC2H3O2) and sodium acetate (NaC2H3O2)
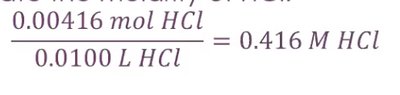
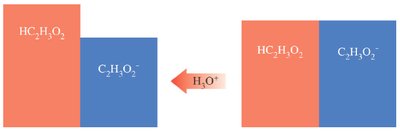
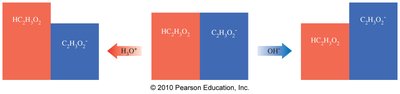
Buffer Function in Health
Buffers are crucial in biological systems, maintaining pH in blood and cells. Blood buffers keep pH close to 7.4, essential for oxygen transport and cellular function.
Summary
Acids and bases are defined by their ability to donate or accept protons or electron pairs.
Equilibrium and Le Chatelier’s Principle explain how reactions respond to changes.
Strong acids/bases dissociate completely; weak ones only partially.
pH is a logarithmic measure of acidity; buffers maintain pH stability.
Titration is a quantitative method for determining acid/base concentration.
