 Back
BackAcids, Bases, and Equilibrium: Study Guide for College Chemistry
Study Guide - Smart Notes

Acids and Bases: Introduction and Clinical Relevance
Acids and Bases: Definitions and Properties
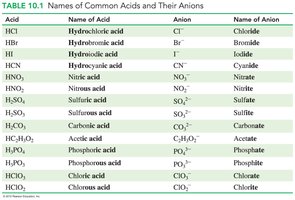
Acids: Characteristics and Naming
Acids are substances that produce ions in water, one of which is the hydronium ion (H3O+). They are classified as electrolytes and are known for their sour taste, ability to turn blue litmus red, and their corrosive properties.
Arrhenius acids: Produce H+ ions in water.
Brønsted–Lowry acids: Donate H+ ions.
Naming acids: Acids with H+ and a nonmetal use the prefix 'hydro' and end with 'ic acid' (e.g., HCl is hydrochloric acid). Acids with H+ and a polyatomic ion change 'ate' to 'ic acid' and 'ite' to 'ous acid'.

Bases: Characteristics and Naming
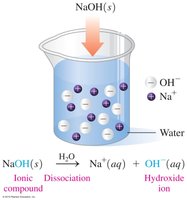
Bases are substances that produce hydroxide ions (OH-) in water. They are also electrolytes, taste bitter or chalky, feel slippery, and turn litmus paper blue.
Arrhenius bases: Produce OH- ions in water.
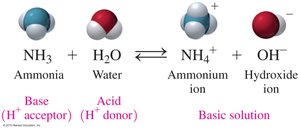
Brønsted–Lowry bases: Accept H+ ions.
Naming bases: Typical bases are named as hydroxides (e.g., NaOH is sodium hydroxide).
Brønsted–Lowry Theory
Acid and Base Reactions
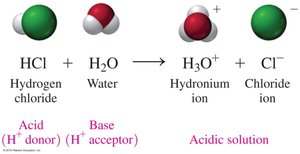
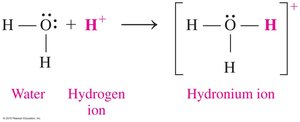
According to the Brønsted–Lowry theory, acids donate H+ ions and bases accept H+ ions. This theory expands the definition of acids and bases beyond those that produce H+ or OH- in water.
Example: HCl + H2O → H3O+ + Cl-
Example: NH3 + H2O → NH4+ + OH-
Characteristics of Acids and Bases
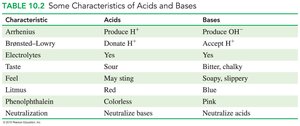
Acids and bases differ in their properties, as summarized below:
Characteristic | Acids | Bases |
|---|---|---|
Arrhenius | Produce H+ | Produce OH- |
Brønsted–Lowry | Donate H+ | Accept H+ |
Electrolytes | Yes | Yes |
Taste | Sour | Bitter, chalky |
Feel | May sting | Soapy, slippery |
Litmus | Red | Blue |
Phenolphthalein | Colorless | Pink |
Neutralization | Neutralize bases | Neutralize acids |
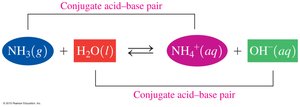
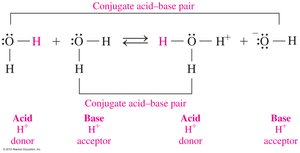
Conjugate Acid–Base Pairs
Definition and Identification
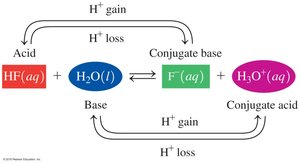
In acid–base reactions, conjugate pairs are formed by the loss and gain of H+. Each acid has a conjugate base, and each base has a conjugate acid.
Example: HF + H2O ⇌ F- + H3O+
Example: NH3 + H2O ⇌ NH4+ + OH-

Strengths of Acids and Bases
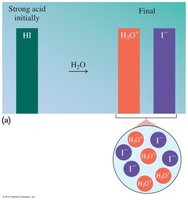
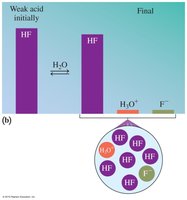
Strong vs. Weak Acids
Strong acids completely ionize in water, while weak acids only partially dissociate. The strength of an acid is determined by its degree of ionization.
Strong acids: HCl, HBr, HI, HNO3, H2SO4, HClO4
Weak acids: HF, H2CO3, CH3COOH




Strong Bases
Strong bases are formed from metals in Groups 1A and 2A and dissociate completely in water.
Examples: NaOH, KOH, Ba(OH)2, Ca(OH)2

Acid–Base Equilibrium
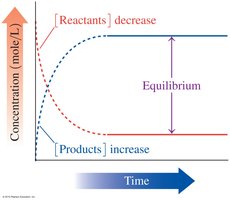
Reversible Reactions and Equilibrium
Acid–base reactions can reach equilibrium, where the rates of the forward and reverse reactions are equal and concentrations remain constant.
Example: HF + H2O ⇌ F- + H3O+

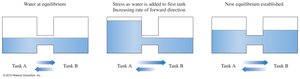
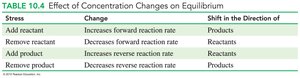
Le Châtelier’s Principle
When a system at equilibrium is disturbed by changing the concentration of reactants or products, the system shifts to relieve the stress and reestablish equilibrium.
Stress | Change | Shift in Direction |
|---|---|---|
Add reactant | Increases forward reaction rate | Products |
Remove reactant | Decreases forward reaction rate | Reactants |
Add product | Increases reverse reaction rate | Reactants |
Remove product | Decreases reverse reaction rate | Products |
Dissociation of Water and Ion Product Constant
Ionization of Water
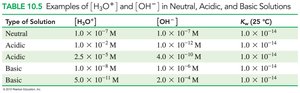
Water is amphoteric and can act as both an acid and a base. The dissociation of water produces equal concentrations of H3O+ and OH- ions at 25°C.
Equation:
Ion product constant: at 25°C
Acidic, Basic, and Neutral Solutions
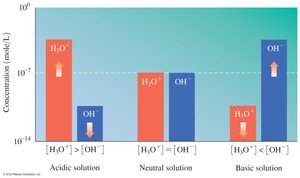
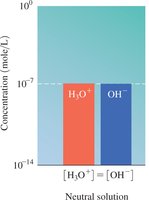
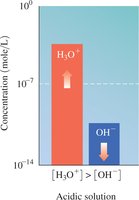
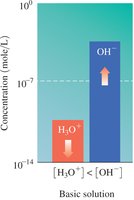
The relationship between [H3O+] and [OH-] determines whether a solution is acidic, basic, or neutral.
Acidic: [H3O+] > [OH-]
Basic: [H3O+] < [OH-]
Neutral: [H3O+] = [OH-]
![Guide to calculating [H3O+] and [OH-]](https://static.studychannel.pearsonprd.tech/study_guide_files/gob/sub_images/f44db6c7_image_26.png)
![Comparison of [H3O+] and [OH-]](https://static.studychannel.pearsonprd.tech/study_guide_files/gob/sub_images/f44db6c7_image_30.png)
![Guide to calculating [H3O+] and [OH-]](https://static.studychannel.pearsonprd.tech/study_guide_files/gob/sub_images/f44db6c7_image_32.png)
The pH Scale
Definition and Calculation
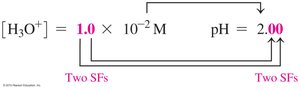
The pH scale measures the acidity of a solution, ranging from 0 (most acidic) to 14 (most basic). pH is calculated as the negative logarithm of the hydronium ion concentration:
Formula:
Acidic: pH < 7
Neutral: pH = 7
Basic: pH > 7

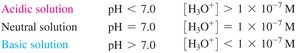


Calculating [H3O+] from pH
Formula:
![Guide to calculating [H3O+] from pH](https://static.studychannel.pearsonprd.tech/study_guide_files/gob/sub_images/f44db6c7_image_44.png)
Reactions of Acids and Bases
Acids Reacting with Metals, Carbonates, and Bases
Acids react with metals to produce hydrogen gas and a salt, with carbonates and bicarbonates to produce carbon dioxide, salt, and water, and with bases in neutralization reactions to produce salt and water.
Example:
Example:
Neutralization:



Acid–Base Titration
Laboratory Procedure and Calculations
Titration is used to determine the molarity of an acid or base by neutralizing a measured volume with a known concentration of the other. The endpoint is identified using an indicator such as phenolphthalein.
Calculation steps: State quantities, write a plan, state equalities and conversion factors, set up the calculation.

Buffers
Definition and Function
A buffer solution maintains pH by neutralizing small amounts of added acid or base. It contains a weak acid and its conjugate base (or a weak base and its conjugate acid).
Example: Acetic acid (HC2H3O2) and sodium acetate (NaC2H3O2)
Function: The weak acid neutralizes added base, and the conjugate base neutralizes added acid.
