 Back
BackAmino Acids and Proteins: Structure, Properties, and Functions
Study Guide - Smart Notes

Chapter 18: Amino Acids and Proteins
18.1 An Introduction to Biochemistry
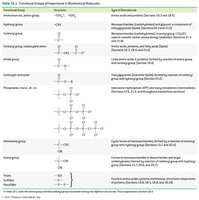
Biochemistry is the study of molecules and their reactions in living organisms. It is foundational to understanding health, disease, nutrition, and pharmaceutical development. Biochemistry integrates principles from inorganic and organic chemistry to explain the structure and function of biomolecules.
Key biomolecules: Proteins, carbohydrates, lipids, nucleic acids
Biochemical reactions: Breakdown of food, energy generation/storage, biosynthesis, waste elimination
Functional groups: Biomolecules contain functional groups similar to those in simpler organic molecules
18.2 Proteins and Their Functions: An Overview
Proteins are essential biomolecules, comprising about 50% of the dry weight of the human body. Their name derives from the Greek 'proteios', meaning 'primary', reflecting their fundamental importance.
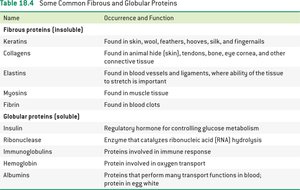
Structural proteins: Provide support (e.g., keratin, actin)
Enzymes: Catalyze biochemical reactions (e.g., catalase)
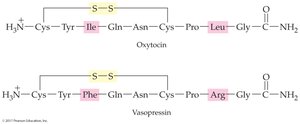
Hormones: Regulate metabolism (e.g., oxytocin)
Transport proteins: Move substances (e.g., transferrin)
Storage proteins: Store molecules (e.g., casein)
Immune proteins: Protect against pathogens (e.g., Immunoglobulin G)
Shape and function: The three-dimensional shape of a protein is critical to its function
Type | Function | Example |
|---|---|---|
Enzymes | Catalyze biochemical reactions | Amylase, trypsin |
Storage proteins | Store nutrients | Casein, ferritin |
Hormones | Regulate body functions | Insulin, oxytocin |
Transport proteins | Carry substances | Transferrin, hemoglobin |
Structural proteins | Support tissues | Collagen, keratin |
Protective proteins | Defend body | Immunoglobulins |
Contractile proteins | Do mechanical work | Myosin |

18.3 Amino Acids
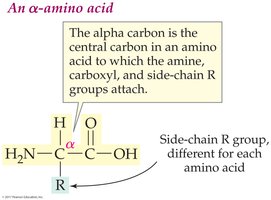
Amino acids are the building blocks of proteins. Each amino acid contains an amine group (–NH2), a carboxyl group (–COOH), and a unique side chain (R group) attached to a central alpha carbon.
Alpha-amino acids: Amine group attached to the alpha carbon
R group: Determines the identity and properties of each amino acid
20 standard amino acids: Used in protein synthesis
Classification: Neutral, acidic, or basic; neutral further divided into nonpolar (hydrophobic) and polar (hydrophilic)
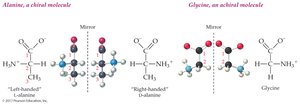
Chirality: 19 amino acids are chiral (except glycine); only L-enantiomers are used in proteins
Intermolecular Forces in Amino Acids
Hydrogen bonding
Van der Waals forces
Ionic bonding
Disulfide bonds
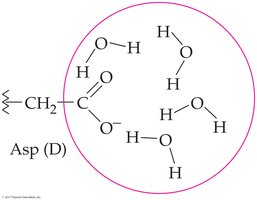
Hydrophobic side chains cluster to avoid water, while hydrophilic side chains interact with water, imparting solubility.
18.4 Acid-Base Properties of Amino Acids
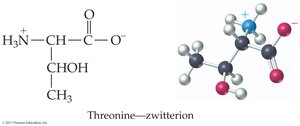
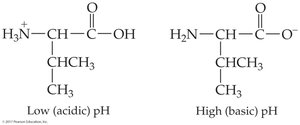
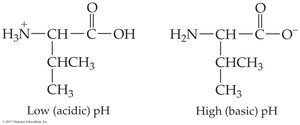
Amino acids contain both acidic (–COOH) and basic (–NH2) groups, allowing them to act as acids and bases. They can form zwitterions, which are dipolar ions with both positive and negative charges.
Zwitterion: Formed by intramolecular acid-base reaction
Physical properties: Crystalline, high melting points, water soluble
pH effects: At low pH, amino acids are positively charged; at high pH, negatively charged
Isoelectric point (pI): pH at which net charge is zero; varies by amino acid
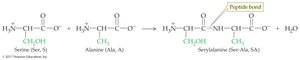
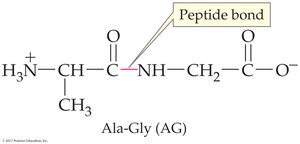
18.5 Peptides
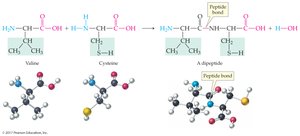
Peptides are chains of amino acids linked by peptide bonds (amide bonds). The sequence of amino acids determines the identity and function of the peptide or protein.
Dipeptide: Two amino acids joined by a peptide bond
Tripeptide: Three amino acids joined
Polypeptide: Many amino acids joined; large peptides are proteins
N-terminal: Free –NH3+ group (left)
C-terminal: Free –COO– group (right)
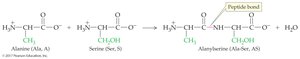
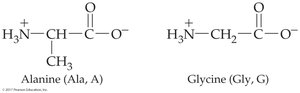
18.6 Protein Structure: Primary Structure (1°)
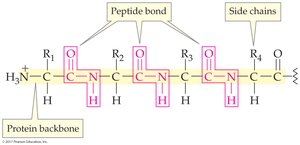
The primary structure of a protein is the sequence of amino acids in its polypeptide chain. This sequence is crucial for the protein's function; even a single amino acid change can alter biological activity.
Backbone: Alternating peptide bonds and alpha-carbon atoms
Side chains: Attached to alpha-carbon atoms
Planar units: Backbone atoms lie in a zigzag, planar arrangement
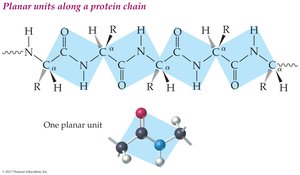
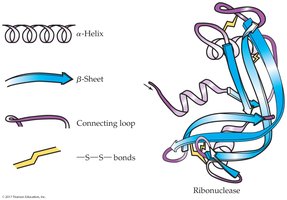
18.7 Secondary Protein Structure (2°)
Secondary structure refers to the spatial arrangement of the polypeptide backbone, stabilized by hydrogen bonding. The two main types are alpha-helix and beta-sheet.
Alpha-helix: Right-handed coil stabilized by hydrogen bonds between backbone atoms
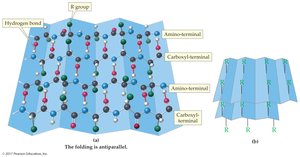
Beta-sheet: Flat sheet-like structure formed by hydrogen bonds between adjacent chains
Fibrous proteins: Tough, insoluble, form fibers or sheets
Globular proteins: Water-soluble, compact shape
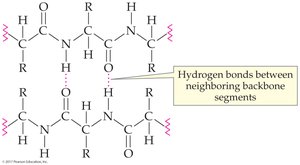
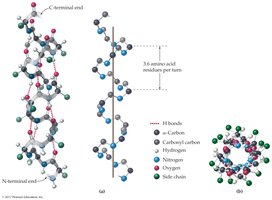

18.8 Tertiary Protein Structure (3°)
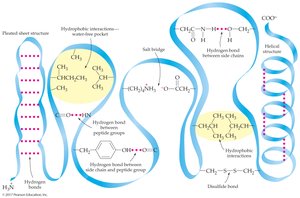
Tertiary structure is the overall three-dimensional folding of a protein, determined by interactions between side chains. Simple proteins contain only amino acid residues; conjugated proteins include non-amino acid units.
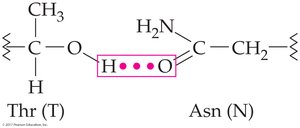
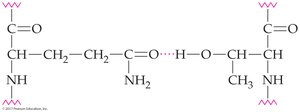
Hydrogen bonds: Between R groups or backbone atoms
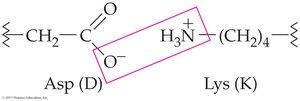
Ionic attractions (salt bridges): Between ionized acidic and basic side chains
Hydrophilic interactions: Between charged R groups and water
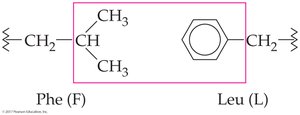
Hydrophobic interactions: Between hydrocarbon side chains
Disulfide bonds: Covalent S–S bonds between cysteine residues
Class of Protein | Nonprotein Part | Examples |
|---|---|---|
Glycoproteins | Carbohydrates | Cell membranes |
Lipoproteins | Lipids | Transport cholesterol |
Metalloproteins | Metal ions | Cytochrome oxidase |
Phosphoproteins | Phosphate groups | Milk casein |
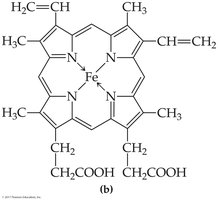
Hemoproteins | Heme | Hemoglobin, myoglobin |
Nucleoproteins | RNA | Ribosomes |


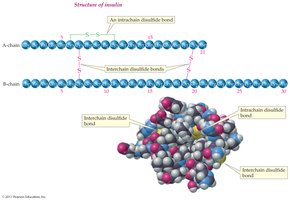
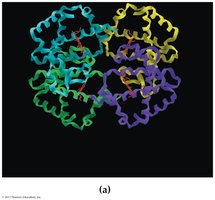
18.9 Quaternary Protein Structure (4°)
Quaternary structure describes the aggregation of two or more polypeptide chains into a larger, ordered structure. These are stabilized by noncovalent forces and sometimes covalent bonds or non-amino acid portions.
Hemoglobin: Four polypeptide chains, four heme groups, carries oxygen in blood
Collagen: Three intertwined chains, major constituent of connective tissues
Serum albumin: Mobile protein in extracellular fluid
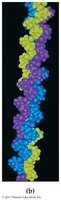
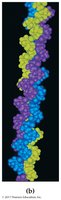
Summary of Protein Structure
Primary: Sequence of amino acids (peptide bonds)
Secondary: Alpha-helices and beta-sheets (hydrogen bonds)
Tertiary: Three-dimensional folding (hydrophilic/hydrophobic interactions, salt bridges, hydrogen bonds, disulfide bonds)
Quaternary: Assembly of multiple polypeptide chains (same interactions as tertiary)
18.10 Chemical Properties of Proteins
Proteins can be hydrolyzed chemically or enzymatically to yield amino acids. Denaturation is the loss of secondary, tertiary, and quaternary structure, often resulting in loss of function.
Hydrolysis: Peptide bonds broken by water or enzymes
Denaturation: Loss of higher-order structure; caused by heat, agitation, detergents, organic compounds, pH changes, inorganic salts
Renaturation: Recovery of biological activity if structure is restored
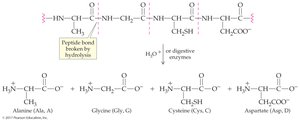
Example: Chymotrypsin hydrolyzes peptide bonds on the carboxyl-terminal side of aromatic amino acids, producing fragments from vasopressin.
Agents of denaturation:
Heat
Mechanical agitation
Detergents
Organic compounds
pH change
Inorganic salts
Additional info: All information needed for protein folding is present in the primary structure. Misfolding leads to loss of function and disease.
