Back
BackAtoms and Elements: Structure, Properties, and Periodic Trends
Study Guide - Smart Notes

Chapter 4: Atoms and Elements
Introduction
This chapter explores the fundamental building blocks of matter—atoms and elements. It covers the classification of matter, the structure of atoms, the organization of the periodic table, and the trends in elemental properties. Understanding these concepts is essential for further study in chemistry and its applications in fields such as agriculture, health, and materials science.
Classification of Matter
Pure Substances and Mixtures
Pure substances are materials made of only one type of particle; they include elements and compounds.
Mixtures contain two or more substances physically combined and can be separated by physical means.
Elements are pure substances from which all other things are built and cannot be broken down into simpler substances.
Elements and Chemical Symbols
Element Names and Symbols
Each element is represented by a unique chemical symbol, usually derived from its English or Latin name. Symbols consist of one or two letters, with the first letter always capitalized.
Examples: C (carbon), N (nitrogen), F (fluorine), Co (cobalt), Ca (calcium)
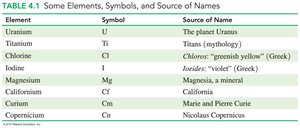
Some symbols are based on Latin names, such as Ag (argentum for silver) and Au (aurum for gold).


Examples of Elements
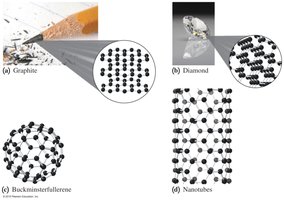
Carbon (C): Exists in several forms, including graphite, diamond, buckminsterfullerene, and nanotubes.
Mercury (Hg): Chemistry Link to Health
Mercury is a liquid metal at room temperature and can be toxic to humans through inhalation, skin contact, or ingestion.
Long-term exposure can damage the brain and kidneys.

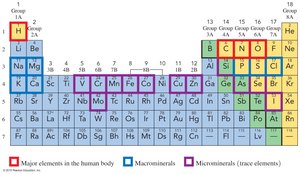
The Periodic Table
Organization of the Periodic Table
The periodic table arranges 118 elements into groups (vertical columns) and periods (horizontal rows) based on similar properties and increasing atomic number.
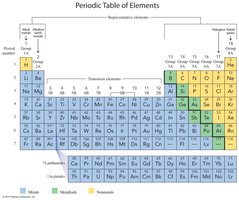
Groups and Periods
Groups: Vertical columns with similar chemical properties (numbered 1A–8A for representative elements, 1–18 in the IUPAC system).
Periods: Horizontal rows numbered 1–7.
Special Groups
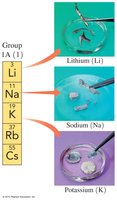
Alkali Metals (Group 1A): Highly reactive metals including Li, Na, K, Rb, Cs.
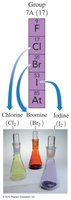
Halogens (Group 7A): Reactive nonmetals including F, Cl, Br, I, At.
Elements Essential for Life
20 elements are essential for human health; O, C, H, and N make up 96% of body mass.
Macrominerals (e.g., Ca, P, K, Cl, S, Na, Mg) are vital for physiological functions.
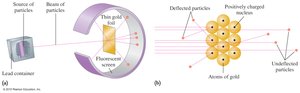
The Structure of the Atom
Dalton’s Atomic Theory
Atoms are tiny particles of matter, unique to each element.
Atoms combine to form compounds and are rearranged in chemical reactions but are not created or destroyed.
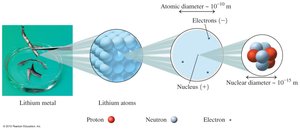
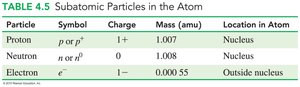
Subatomic Particles
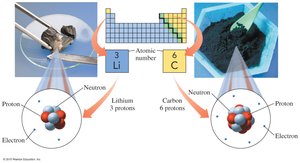
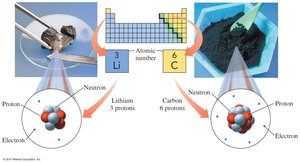
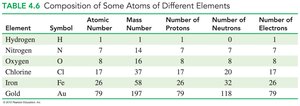
Protons: Positive charge, located in the nucleus, mass ≈ 1 amu.
Neutrons: No charge, located in the nucleus, mass ≈ 1 amu.
Electrons: Negative charge, located outside the nucleus, mass ≈ 0.00055 amu.

Atomic Number and Mass Number
Atomic Number (Z)
The atomic number is the number of protons in an atom and defines the element.
For a neutral atom, the number of electrons equals the number of protons.
Mass Number (A)
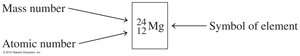
The mass number is the sum of protons and neutrons in the nucleus.
Number of neutrons = mass number – atomic number.
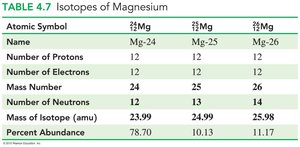
Isotopes and Atomic Mass
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons and thus different mass numbers.
Isotopes are represented by their atomic symbol, mass number, and atomic number.

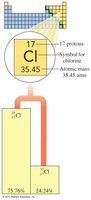
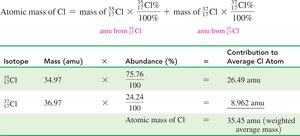
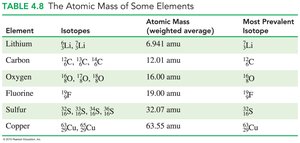
Calculating Atomic Mass
The atomic mass of an element is the weighted average of the masses of its naturally occurring isotopes.
Atomic mass = (mass of isotope 1 × percent abundance) + (mass of isotope 2 × percent abundance) + ...
Electron Energy Levels
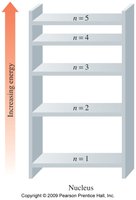
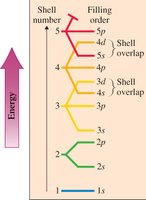
Energy Levels (Shells)
Electrons occupy energy levels (shells) designated by n = 1, 2, 3, ...
Each shell can hold a maximum number of electrons:
Subshells and Orbitals
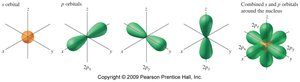
Each shell contains subshells (s, p, d, f) with specific shapes and capacities.
Each orbital can hold up to 2 electrons with opposite spins.
Electron Configurations
Electron configuration describes the arrangement of electrons in shells and subshells.
Example: Oxygen (O): 1s22s22p4
Trends in Periodic Properties
Valence Electrons and Electron-Dot Symbols
Valence electrons are the electrons in the outermost shell and determine chemical properties.
Electron-dot symbols (Lewis structures) represent valence electrons as dots around the element symbol.
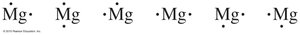


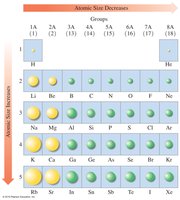
Atomic Size
Atomic size increases down a group and decreases across a period from left to right.
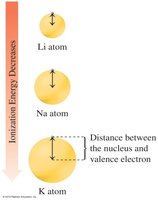
Ionization Energy
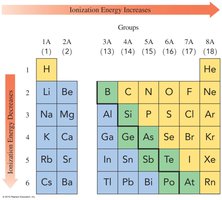
Ionization energy is the energy required to remove an electron from an atom.
Ionization energy decreases down a group and increases across a period.
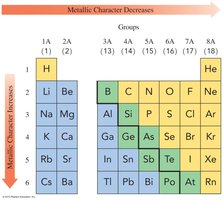
Metallic Character
Metallic character refers to the tendency to lose electrons and form positive ions.
Metallic character increases down a group and decreases across a period.
Summary of Periodic Trends
Periodic Property | Top to Bottom (Group) | Left to Right (Period) |
|---|---|---|
Valence Electrons | Remains the same | Increases |
Atomic Size | Increases | Decreases |
Ionization Energy | Decreases | Increases |
Metallic Character | Increases | Decreases |

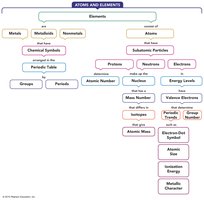
Concept Map
The following concept map summarizes the relationships among the main ideas in this chapter, including the classification of elements, atomic structure, and periodic trends.