Back
BackChapter 16: Amines – Structure, Properties, and Biological Significance
Study Guide - Smart Notes

Chapter 16: Amines
16.1 Classifying Amines
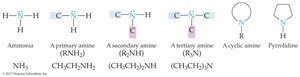
Amines are organic compounds derived from ammonia (NH3), in which one or more hydrogen atoms are replaced by organic groups. The classification of amines is based on the number of organic groups attached to the nitrogen atom.
Primary amine (1°): One organic group attached to nitrogen (RNH2).
Secondary amine (2°): Two organic groups attached to nitrogen (R2NH).
Tertiary amine (3°): Three organic groups attached to nitrogen (R3N).
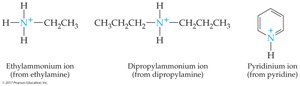
Quaternary ammonium ion: Four organic groups attached to nitrogen, resulting in a positively charged ion (R4N+).
Cyclic amines: Organic groups may be connected in a ring structure, forming heterocyclic amines.
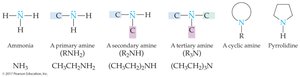
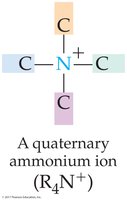
Key Point: The lone pair of electrons on the nitrogen atom is central to amine chemistry, allowing for the formation of quaternary ammonium ions and influencing basicity.
16.2 Naming and Drawing Amines
The nomenclature of amines depends on their classification and structure. The naming conventions are as follows:
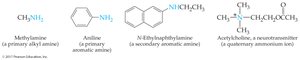
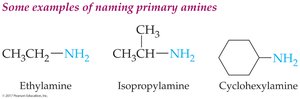
Primary alkyl amines: Name the alkyl group and add the suffix "-amine."
Secondary and tertiary amines (simple): Use "di-" or "tri-" prefixes with the alkyl group name and add "-amine."
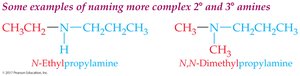
Secondary and tertiary amines (complex): Name as N-substituted derivatives of the largest parent group; other groups are N-substituents.
Amino group: The –NH2 group is called an amino group; "amino-" is used as a prefix when it is a substituent.
Aromatic amines: Often known by common names, e.g., aniline.
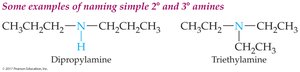

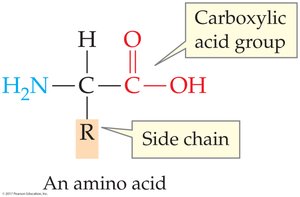
Proteins are polymers of amino acids, which have the general structure H2N—CHR—COOH.
Worked Example 16.1: N,N-diethylbutylamine
Analysis: The parent compound is butylamine; N,N indicates two ethyl groups bonded to nitrogen. Solution: Three alkyl groups are bonded to N, so it is a tertiary amine.
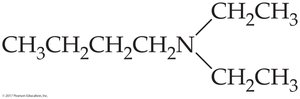
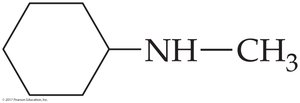
Worked Example 16.2: N-methylcyclohexylamine
Analysis: Two groups (cyclohexyl and methyl) are bonded to N. Solution: It is a secondary amine.
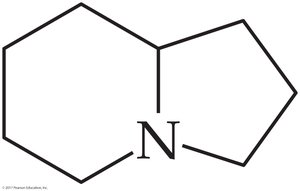
Worked Example 16.3: Octahydroindolizine
Analysis: Nitrogen is attached to three carbons in a ring. Solution: It is a tertiary amine.
16.3 Properties of Amines
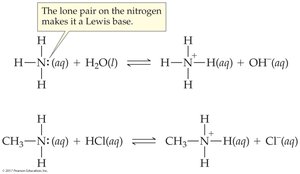
Amines exhibit characteristic physical and chemical properties due to the lone pair of electrons on nitrogen. They act as weak Brønsted-Lowry bases or Lewis bases.
Lewis base: Compound with an unshared pair of electrons.
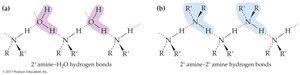
Hydrogen bonding: Primary and secondary amines can hydrogen bond with each other and with water.
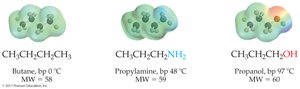
Boiling points: Primary and secondary amines have higher boiling points than alkanes of similar size; alcohols have even higher boiling points due to more hydrogen bonding.
Solubility: Amines with up to six carbon atoms are appreciably soluble in water.
Odor and toxicity: Many amines have strong odors and can be toxic or irritating.
16.4 Heterocyclic Nitrogen Compounds
Heterocyclic compounds contain nitrogen atoms within a ring structure. These may be aromatic or nonaromatic and are common in biological molecules and drugs.
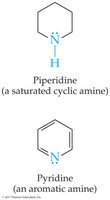
Examples: Piperidine (saturated cyclic amine), Pyridine (aromatic amine).
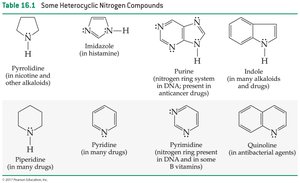
Compound | Structure | Notes |
|---|---|---|
Pyrrolidine | Ring with N | In nicotine and alkaloids |
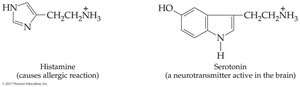
Imidazole | Ring with two N | In histamine |
Purine | Double ring with N | In DNA, anticancer drugs |
Indole | Double ring with N | In alkaloids and drugs |
Piperidine | Ring with N | In drugs |
Pyridine | Ring with N | In drugs |
Pyrimidine | Ring with N | In DNA, B vitamins |
Quinoline | Double ring with N | Antibacterial agents |
16.5 Basicity of Amines
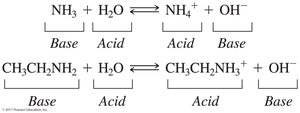
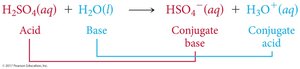
Amines are weak bases in aqueous solution, forming OH– and ammonium ions. Their basicity depends on their structure.
Reaction with water: Amines accept a hydrogen ion from water, forming an ammonium ion and hydroxide ion.
Reaction with acids: Amines react with hydronium ions to form ammonium ions and water.
Naming: Alkylamine cations are named by replacing "-amine" with "-ammonium."
Acidity: Ammonium ions with at least one hydrogen are weakly acidic and can react with bases to regenerate the amine.
Biological context: Amines exist as ammonium ions in blood and body fluids.
Basicity comparison: Nonaromatic amines > Ammonia > Aromatic amines.
Example Equations:
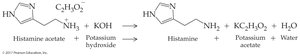
Worked Example 16.5: Histamine acetate reacts with KOH to regenerate the free amine, water, and potassium acetate.
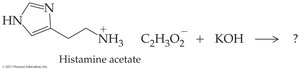
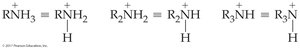
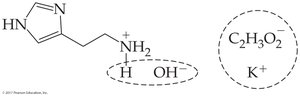
16.6 Amine Salts
Ammonium salts are ionic compounds formed from amines and acids. They are generally odorless, white, crystalline solids with high solubility.
Naming: Combine the names of the amine and the acid (e.g., methylamine hydrochloride).
Regeneration: Free amines can be regenerated from their salts by treatment with a base.
Quaternary ammonium ions: Have four organic groups, are permanently charged, and are not affected by pH changes.
Medicinal use: Benzalkonium chlorides are used as antiseptics and disinfectants.


16.7 Amines in Plants: Alkaloids
Alkaloids are naturally occurring, nitrogen-containing compounds isolated from plants. They are typically basic, bitter, and often poisonous.
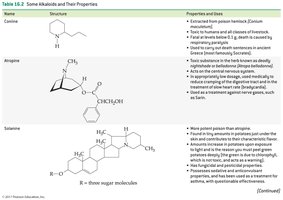
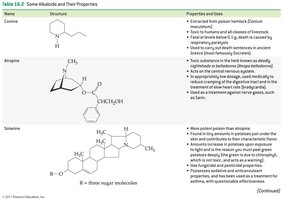
Coniine: Extracted from poison hemlock; toxic to humans and animals.
Atropine: Found in deadly nightshade; acts on the central nervous system and used in medicine.
Solanine: Found in potatoes and tomatoes; production increases with sunlight or improper storage.
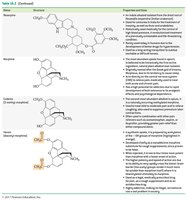
Morphine and codeine: Present in poppy; used medicinally for pain relief.
Heroin: Synthesized from morphine; converted back to morphine in the body.
Name | Structure | Properties and Uses |
|---|---|---|
Coniine | Structure shown | Poison hemlock; toxic, used historically for executions |
Atropine | Structure shown | Deadly nightshade; CNS effects, used in medicine |
Solanine | Structure shown | Potatoes/tomatoes; toxic, increased by sunlight |
Morphine | Structure shown | Poppy; pain relief, medicinal use |
Codeine | Structure shown | Poppy; pain relief, medicinal use |
Heroin | Structure shown | Synthesized from morphine; converted in body |