 Back
BackChapter 6: Ionic and Molecular Compounds – Study Notes
Study Guide - Smart Notes

Chapter 6: Ionic and Molecular Compounds
Introduction to Ionic and Molecular Compounds
Chemical compounds are classified into two main types: ionic compounds and molecular compounds. These classes differ in their bonding, properties, and the types of elements involved.
Ionic compounds typically have high melting points (500°C – 2000°C) and conduct electricity when molten or dissolved in water.
Molecular compounds generally have lower melting points and may exist as gases or liquids at room temperature.
6.1 Ions: Transfer of Electrons
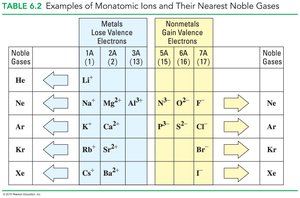
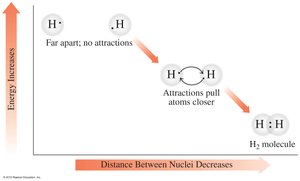
Atoms form ions by losing or gaining electrons to achieve a stable electron configuration, often resembling the nearest noble gas. The resulting ions are held together by ionic bonds, which are strong electrostatic attractions between oppositely charged ions.
Cations are positively charged ions formed when metals lose electrons.
Anions are negatively charged ions formed when nonmetals gain electrons.
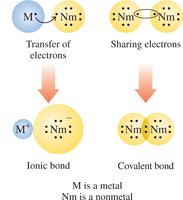
Formation of Positive Ions (Cations)
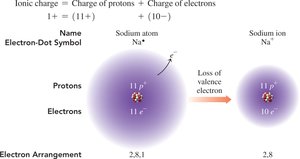
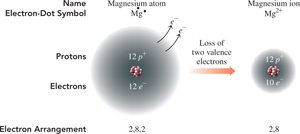
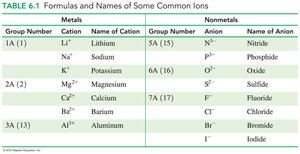
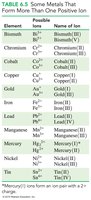
Metals in Groups 1A, 2A, and 3A lose electrons to form cations with charges equal to their group number.
Example: Sodium (Na) loses one electron to form Na+.
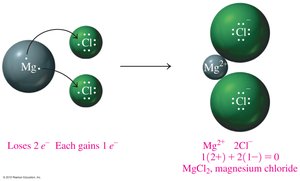
Example: Magnesium (Mg) loses two electrons to form Mg2+.
Formation of Negative Ions (Anions)
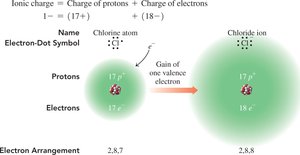
Nonmetals in Groups 5A, 6A, and 7A gain electrons to form anions with charges equal to 8 minus their group number.
Example: Chlorine (Cl) gains one electron to form Cl−.
Common Ions and Their Charges
The periodic table group numbers help predict the charges of representative element ions.
6.2 Writing Formulas for Ionic Compounds
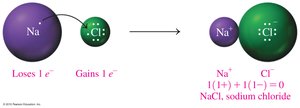
Ionic compounds are composed of cations and anions in ratios that result in overall electrical neutrality. The chemical formula reflects the lowest whole-number ratio of ions.
The sum of positive and negative charges must equal zero:
Subscripts indicate the number of each ion needed for charge balance.
Examples of Ionic Compound Formation
Na+ and Cl− combine to form NaCl.
Mg2+ and Cl− combine to form MgCl2.
6.3 Naming Ionic Compounds
The name of an ionic compound consists of the cation (metal) name followed by the anion (nonmetal) name with an -ide ending. For metals with variable charges (mainly transition metals), a Roman numeral indicates the charge.
Example: NaCl is named sodium chloride.
Example: FeCl2 is named iron(II) chloride.

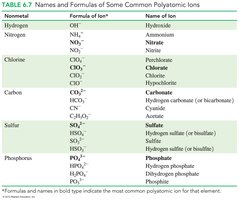
6.4 Polyatomic Ions
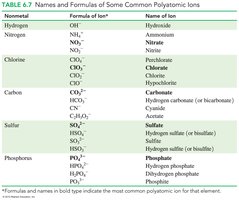
Polyatomic ions are groups of covalently bonded atoms that carry an overall charge. Most are negatively charged, except for ammonium (NH4+).
Examples include hydroxide (OH−), nitrate (NO3−), sulfate (SO42−), and carbonate (CO32−).
When writing formulas, use parentheses if more than one polyatomic ion is needed.

6.5 Molecular Compounds: Sharing Electrons
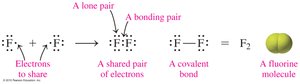
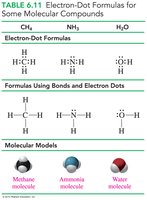
Molecular compounds are formed when nonmetal atoms share electrons, creating covalent bonds. The number of bonds typically equals the number of electrons needed to complete an octet.
Examples: H2O (water), NH3 (ammonia), CH4 (methane).
Some elements exist as diatomic molecules (e.g., H2, O2, N2).
Multiple Bonds
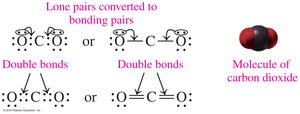
Double bonds involve sharing two pairs of electrons (e.g., O2, CO2).
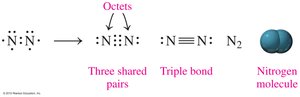
Triple bonds involve sharing three pairs of electrons (e.g., N2).
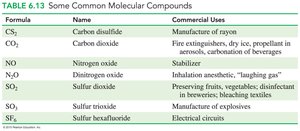
Naming Molecular Compounds
The first nonmetal is named by its element name.
The second nonmetal is named with the -ide ending.
Prefixes indicate the number of each atom (e.g., CO2: carbon dioxide).

6.6 Electronegativity and Bond Polarity
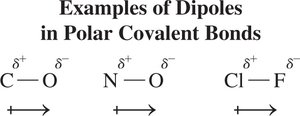
Electronegativity is a measure of an atom's ability to attract shared electrons. The difference in electronegativity between two atoms determines the bond type:
Nonpolar covalent: difference 0–0.4
Polar covalent: difference 0.5–1.8
Ionic: difference >1.8
6.7 Shapes and Polarity of Molecules
The valence shell electron-pair repulsion (VSEPR) theory predicts the shapes of molecules based on the repulsion between electron groups around a central atom. Common shapes include linear, trigonal planar, tetrahedral, trigonal pyramidal, and bent.
Nonpolar molecules have symmetrical shapes or nonpolar bonds.
Polar molecules have asymmetrical shapes or polar bonds that do not cancel.
6.8 Attractive Forces in Compounds
Attractive forces between particles determine the physical properties of substances:
Ion-ion interactions: Strongest, occur in ionic compounds.
Dipole-dipole attractions: Occur between polar molecules.
Hydrogen bonds: Strong dipole-dipole attractions involving H bonded to F, O, or N.
Dispersion forces: Weakest, present in all molecules, especially nonpolar ones.
The strength of these forces affects melting and boiling points.
Summary Table: Types of Bonds and Forces
Type | Bond/Force | Relative Strength | Example |
|---|---|---|---|
Ionic | Ion-ion | Strongest | NaCl |
Covalent (polar) | Hydrogen bond | Strong | H2O |
Covalent (polar) | Dipole-dipole | Moderate | NCl3 |
Covalent (nonpolar) | Dispersion | Weakest | Br2 |
Additional info: This summary includes expanded academic context, definitions, and examples to ensure completeness and clarity for exam preparation.
