 Back
BackChapter 6: Ionic and Molecular Compounds – Structured Study Notes
Study Guide - Smart Notes

Chapter 6: Ionic and Molecular Compounds
Ionic and Covalent Bonds
Chemical bonds are formed when atoms lose, gain, or share valence electrons to achieve a stable octet (eight valence electrons), known as the octet rule. There are two primary types of chemical bonds:
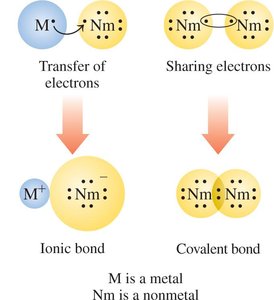
Ionic bonds: Occur when atoms of one element (usually metals) lose valence electrons and atoms of another element (usually nonmetals) gain valence electrons, resulting in the formation of ions.
Covalent bonds: Occur when nonmetal atoms share electrons to attain a noble gas arrangement.
Particles and Bonds in Compounds
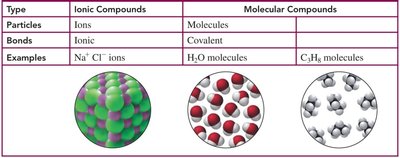
Ionic compounds and molecular compounds differ in the types of particles and bonds they contain:
Type | Ionic Compounds | Molecular Compounds |
|---|---|---|
Particles | Ions | Molecules |
Bonds | Ionic | Covalent |
Examples | Na+, Cl- ions | H2O molecules, C2H6 molecules |
Ions: Transfer of Electrons
Atoms form ions by losing or gaining electrons:
Positive ions (cations): Formed when metals lose electrons.
Negative ions (anions): Formed when nonmetals gain electrons.
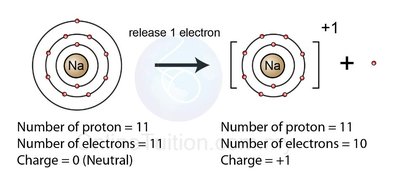
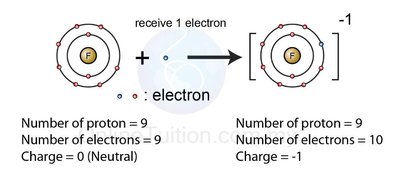
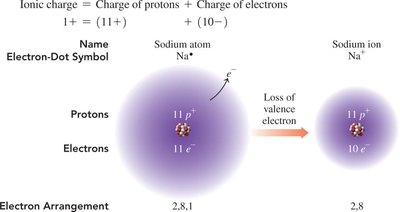
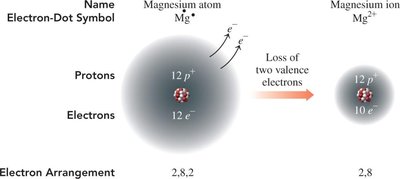
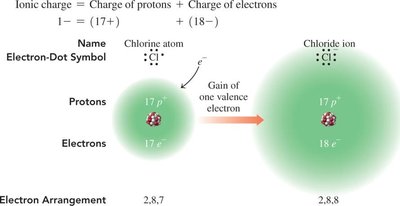
Formation of Ions: Electron Arrangement
When an atom loses or gains electrons, its electron arrangement changes to match that of the nearest noble gas.
Example: Sodium (Na) loses one electron to become Na+ with an electron configuration like neon (Ne).
Example: Magnesium (Mg) loses two electrons to become Mg2+ with an electron configuration like neon (Ne).
Example: Chlorine (Cl) gains one electron to become Cl- with an electron configuration like argon (Ar).
Ionic Charges from Group Numbers
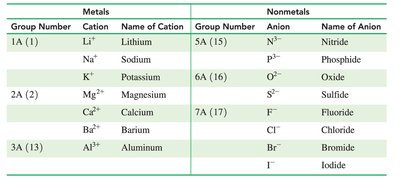
The periodic table group numbers help predict the charges of ions formed by representative elements:
Group Number | Cation/Anion | Name |
|---|---|---|
1A (1) | Li+, Na+, K+ | Lithium, Sodium, Potassium |
2A (2) | Mg2+, Ca2+, Ba2+ | Magnesium, Calcium, Barium |
3A (13) | Al3+ | Aluminum |
5A (15) | N3-, P3- | Nitride, Phosphide |
6A (16) | O2-, S2- | Oxide, Sulfide |
7A (17) | F-, Cl-, Br-, I- | Fluoride, Chloride, Bromide, Iodide |
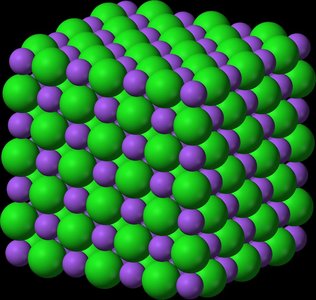
Properties of Ionic Compounds
Ionic compounds consist of positive and negative ions held together by strong ionic bonds. They have:
High melting points
Solid state at room temperature
Electrical conductivity when dissolved in water
Naming and Writing Ionic Formulas
The chemical formula of an ionic compound represents the lowest whole-number ratio of ions, ensuring charge balance:
Total positive charge = total negative charge
Subscripts indicate the number of each ion needed for neutrality
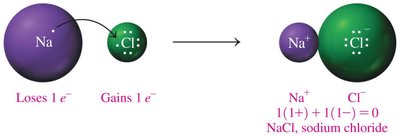
Example: Sodium Chloride Formation
Sodium (Na) loses one electron, chlorine (Cl) gains one electron, forming NaCl:
Na+ + Cl- → NaCl
Charge balance: 1(1+) + 1(1-) = 0
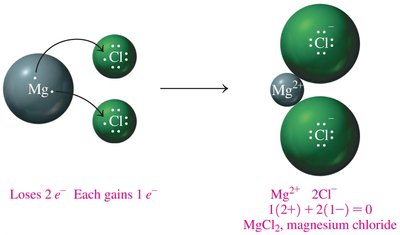
Example: Magnesium Chloride Formation
Magnesium (Mg) loses two electrons, two chlorine (Cl) atoms each gain one electron, forming MgCl2:
Mg2+ + 2Cl- → MgCl2
Charge balance: 1(2+) + 2(1-) = 0
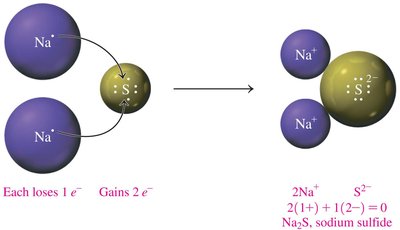
Example: Sodium Sulfide Formation
Two sodium (Na) atoms each lose one electron, sulfur (S) gains two electrons, forming Na2S:
2Na+ + S2- → Na2S
Charge balance: 2(1+) + 1(2-) = 0
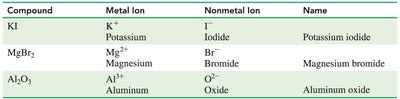
Naming Ionic Compounds
When naming ionic compounds:
The metal (cation) is named first, using its element name.
The nonmetal (anion) is named second, using the stem of its name plus the suffix -ide.
For transition metals with variable charge, a Roman numeral indicates the ion's charge.
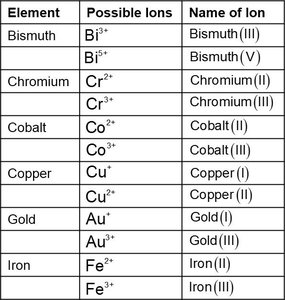
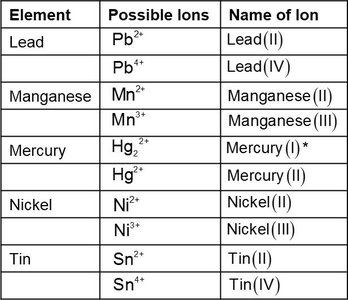
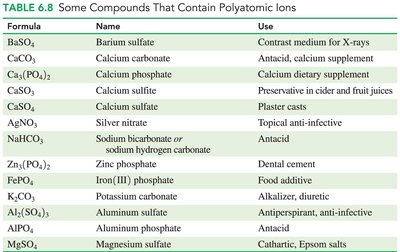
Polyatomic Ions
Polyatomic ions are groups of atoms with an overall charge, often containing oxygen and a nonmetal. Most are negatively charged except ammonium (NH4+).
Names ending in -ate have more oxygen; -ite have one fewer oxygen.
Some exceptions: cyanide (CN-), hydroxide (OH-).
Adding H+ to a polyatomic ion changes its name and charge (e.g., HCO3- is bicarbonate).
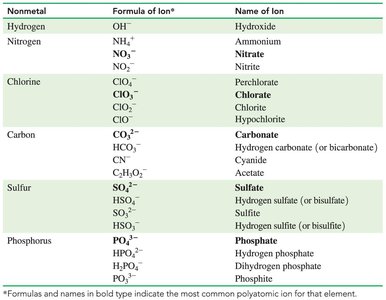
Writing Formulas for Compounds with Polyatomic Ions
To write formulas for compounds containing polyatomic ions:
Identify the cation and polyatomic ion (anion).
Balance the charges.
Write the formula, placing the polyatomic ion in parentheses if more than one is needed.


Guide to Naming Compounds with Polyatomic Ions
Follow these steps:
Identify the cation and polyatomic ion (anion).
Name the cation using a Roman numeral if needed.
Name the polyatomic ion.
Write the name for the compound, cation first and polyatomic ion second.
Molecular Compounds: Sharing Electrons
Molecular compounds form when atoms of two or more nonmetals share electrons, creating covalent bonds. The number of covalent bonds formed is usually equal to the number of electrons needed for a stable arrangement.
Summary Table: Naming Ionic and Molecular Compounds
Key differences in naming and formula writing:
Ionic compounds: Metal + Nonmetal (with -ide ending), charge balance, Roman numerals for variable charge metals.
Molecular compounds: Prefixes indicate number of atoms, both elements are nonmetals, second element ends in -ide.
Additional info:
Polyatomic ions are common in biological and industrial compounds (e.g., plaster casts, fertilizers).
Charge balance is essential for all ionic compounds.
Transition metals often require Roman numerals to specify their charge.
