 Back
BackChapter 8: Gases – Fundamental Properties and Laws
Study Guide - Smart Notes

Gases: Properties and Fundamental Laws
Properties of Gases
Gases are characterized by four main properties: pressure (P), volume (V), temperature (T), and amount (n). These properties are interrelated and form the basis for understanding gas behavior.
Pressure (P): The force exerted by gas particles against the walls of their container. Units include atmosphere (atm), millimeters of mercury (mmHg), torr, and pascal (Pa).
Volume (V): The space occupied by the gas, measured in liters (L), milliliters (mL), or cubic meters (m3).
Temperature (T): Determines the kinetic energy and rate of motion of gas particles. Calculations require temperature in Kelvin (K).
Amount (n): The quantity of gas, typically measured in moles (n).
Kinetic Molecular Theory of Gases
The kinetic molecular theory explains the behavior of ideal gases:
Gases consist of small particles moving rapidly in straight paths.
There are no attractive forces between particles.
Particles are far apart relative to their size.
The kinetic energy of particles increases with temperature (in Kelvin).
Gas Pressure and Measurement
Gas pressure is the force acting on a specific area. Atmospheric pressure decreases at higher altitudes. A barometer is used to measure atmospheric pressure.
1 atm = 760 mmHg = 760 torr = 14.7 lb/in2 = 101.325 kPa
Ideal vs. Real Gases
Ideal gases obey gas laws exactly and are modeled as non-interacting particles. Real gases deviate from ideal behavior due to intermolecular forces, especially at high pressures and low temperatures.
Gas Laws
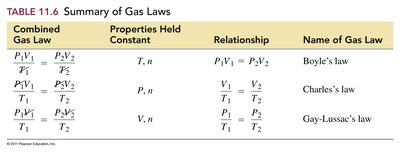
Summary of Gas Laws
The gas laws describe the relationships between pressure, volume, temperature, and amount of gas. The main laws are Boyle's, Charles's, Gay-Lussac's, Avogadro's, and the Combined Gas Law.
Combined Gas Law | Properties Held Constant | Relationship | Name of Gas Law |
|---|---|---|---|
T, n | Boyle's law | ||
P, n | Charles's law | ||
V, n | Gay-Lussac's law |
Boyle's Law: Pressure and Volume
Boyle's law states that the pressure of a gas is inversely related to its volume when temperature and amount are constant.
If pressure increases, volume decreases.
If pressure decreases, volume increases.
Mathematically:
Example: If a sample of Freon gas has an initial volume of 8.0 L at 550 mmHg, and the pressure increases to 2200 mmHg, the new volume is calculated as:
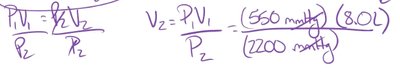
Charles's Law: Volume and Temperature
Charles's law states that the volume of a gas is directly proportional to its Kelvin temperature when pressure and amount are constant.
As temperature increases, volume increases.
Mathematically:

Example: A balloon has a volume of 785 mL at 21°C (294 K). If the temperature drops to 0°C (273 K), the new volume is:
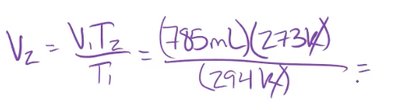
Gay-Lussac's Law: Pressure and Temperature
Gay-Lussac's law states that the pressure of a gas is directly proportional to its Kelvin temperature when volume and amount are constant.
As temperature increases, pressure increases.
Mathematically:
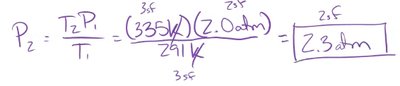
Combined Gas Law
The combined gas law incorporates Boyle's, Charles's, and Gay-Lussac's laws, relating pressure, volume, and temperature when the amount of gas is constant.
Mathematically:
Example: A sample of helium gas has a volume of 0.180 L, a pressure of 0.800 atm, and a temperature of 29°C (302 K). At what temperature will the helium have a volume of 90.0 mL and a pressure of 3.20 atm?
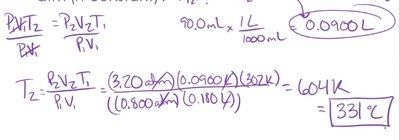
Avogadro's Law: Volume and Moles
Avogadro's law states that the volume of a gas is directly proportional to the number of moles of gas when pressure and temperature are constant.
As the number of moles increases, volume increases.
Mathematically:
Example: If 0.75 mol of helium occupies 1.5 L, what volume will 1.2 mol occupy?
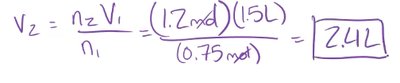
Standard Temperature and Pressure (STP) and Molar Volume
At STP (0°C or 273 K, 1 atm), one mole of an ideal gas occupies 22.4 L. This allows comparison of gas volumes under standard conditions.
The Ideal Gas Law
The ideal gas law relates all four properties of a gas:
P = pressure (atm), V = volume (L), n = moles, T = temperature (K), R = gas constant (0.0821 L·atm/mol·K)
Example: What is the pressure in a 20.0-L tank containing 2.86 mol of N2O at 23°C (296 K)?
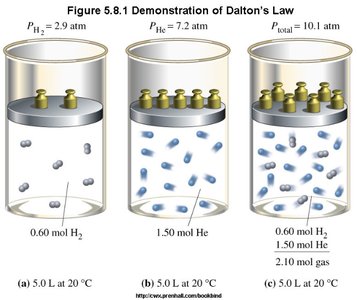
Dalton's Law of Partial Pressures
Dalton's law states that the total pressure of a mixture of gases is the sum of the partial pressures of each gas.
The partial pressure is the pressure each gas would exert if it were alone in the container.
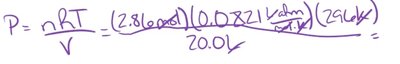
Example: A Heliox mixture for a scuba diver has a total pressure of 7.00 atm. If the partial pressure of oxygen is 1140 mmHg (1.50 atm), what is the partial pressure of helium?
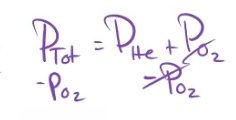
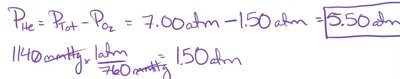
Summary Table: Gas Laws
Law | Relationship | Properties Held Constant | Equation |
|---|---|---|---|
Boyle's Law | Pressure inversely proportional to volume | T, n | |
Charles's Law | Volume directly proportional to temperature | P, n | |
Gay-Lussac's Law | Pressure directly proportional to temperature | V, n | |
Avogadro's Law | Volume directly proportional to moles | P, T | |
Combined Gas Law | Relates P, V, T | n | |
Ideal Gas Law | Relates P, V, n, T | None | |
Dalton's Law | Total pressure is sum of partial pressures | None |
