 Back
BackChapter 9: Solutions – Fundamentals of Chemistry Study Guide
Study Guide - Smart Notes

Solutions
Definition and Components
A solution is a homogeneous mixture composed of two or more substances. It consists of a solvent (the substance present in the greatest amount) and one or more solutes (substances dissolved in the solvent).
Solutes are evenly distributed throughout the solution, cannot be separated by filtration, but can be separated by evaporation.
Solutes are not visible but may impart color to the solution.
Types of Solutions
Solutions can involve solids, liquids, or gases as solutes and solvents. Examples include air (gas in gas), soda water (gas in liquid), seawater (solid in liquid), and brass (solid in solid).
Gas Solutions: Air (O2 in N2), soda water (CO2 in H2O)
Liquid Solutions: Vinegar (acetic acid in water), tincture of iodine (iodine in ethanol)
Solid Solutions: Brass (zinc in copper), steel (carbon in iron)
Water as a Solvent
Water is a common solvent due to its polarity and ability to form hydrogen bonds, allowing it to dissolve ions and polar molecules.
Dissociation and Electrolytes
Dissociation Reactions
When ionic compounds dissolve in water, they dissociate into ions. The (aq) symbol indicates hydrated ions surrounded by water molecules.
Example:
Electrolytes
Electrolytes are substances that produce ions in solution and conduct electricity. They are classified as strong, weak, or nonelectrolytes.
Strong electrolytes: Completely dissociate into ions (e.g., NaCl, CaBr2).
Weak electrolytes: Partially dissociate, producing a few ions (e.g., HF, NH3).
Nonelectrolytes: Dissolve as molecules, do not produce ions, and do not conduct electricity (e.g., sucrose, methanol).
Equivalents
An equivalent (Eq) is the amount of an ion that provides 1 mole of electrical charge.
1 mole Na+ = 1 Eq
1 mole Ca2+ = 2 Eq
1 mole Fe3+ = 3 Eq
Concentration of Solutions
Mass Percent (m/m)
Mass percent is the percent by mass of solute in a solution.
Formula:
Example: 8.00 g KCl in 50.00 g solution gives (m/m)
Volume Percent (v/v)
Volume percent is the percent by volume of solute in a solution.
Formula:
Mass/Volume Percent (m/v)
Mass/volume percent is the percent by mass of solute per volume of solution.
Formula:
Percent Conversion Factors
Percent concentrations can be used as conversion factors in calculations.
Example: 10.0% (m/m) NaCl means 10.0 g NaCl per 100 g solution.
Molarity (M)
Definition and Calculation
Molarity is a concentration term that gives the moles of solute per liter of solution.
Formula:
Example: Preparing 1.0 M NaCl solution by dissolving 58.5 g NaCl (1.00 mole) in 1.0 L water.
Calculating Molarity
To calculate molarity, convert grams of solute to moles, then divide by the volume in liters.
Example: 6.00 g NaOH in 0.500 L solution
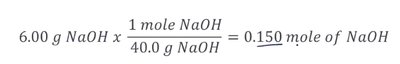

Molarity Conversion Factors
Molarity can be used to convert between volume, moles, and grams of solute.
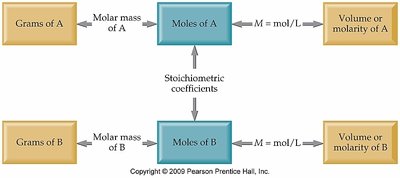
Example: Calculating grams of KCl needed for a specific molarity and volume.

Solubility and Types of Solutions
Solubility
Solubility is the maximum amount of solute that dissolves in a specific amount of solvent, usually expressed as grams per 100 g of water.
Unsaturated solutions: Less than maximum solute, can dissolve more.
Saturated solutions: Maximum solute dissolved, undissolved solute may be present.
Supersaturated solutions: More than maximum solute, unstable.
Effect of Temperature and Pressure
Solubility of most solids increases with temperature; solubility of gases decreases.
Henry’s law: Solubility of a gas in a liquid increases with pressure.
Solubility Rules
Soluble salts contain ions from Group 1A, NH4+, NO3-, or C2H3O2-. Most other combinations are insoluble.
Dilution
Principles of Dilution
Dilution involves adding water to a solution, increasing volume and decreasing concentration. The moles of solute remain constant.
For percent concentration:
For molarity:
Properties of Solutions, Colloids, and Suspensions
Comparison
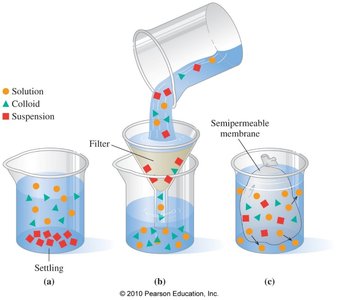
Solutions, colloids, and suspensions differ in particle size, settling, and separation methods.
Solutions: Small particles, do not settle, cannot be separated by filtration or semipermeable membranes.
Colloids: Medium particles, do not settle, can be separated by semipermeable membranes, scatter light.
Suspensions: Large particles, settle out, can be separated by filtration.
Colligative Properties
Freezing Point Depression and Boiling Point Elevation
Colligative properties are changes in solvent properties due to solute particles. Adding solute lowers the freezing point and raises the boiling point.
1 mole of solute in 1000 g water decreases freezing point by 1.86°C and increases boiling point by 0.52°C.
Electrolytes produce more particles, causing greater changes.
Osmosis and Osmotic Pressure
Osmosis
Osmosis is the flow of water from lower to higher solute concentration across a semipermeable membrane. The solution with higher solute concentration rises in volume.
Osmotic Pressure
Osmotic pressure is the pressure required to prevent water flow into a more concentrated solution. It increases with the number of dissolved particles.
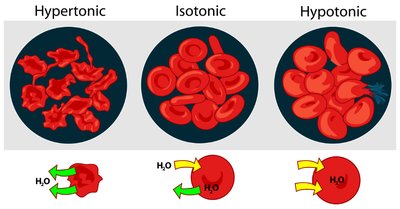
Tonicity: Isotonic, Hypotonic, and Hypertonic Solutions
Red blood cells are affected by the osmotic pressure of their environment:
Isotonic: Equal osmotic pressure; cells retain normal shape.
Hypotonic: Lower osmotic pressure; water enters cells, causing swelling (hemolysis).
Hypertonic: Higher osmotic pressure; water leaves cells, causing shrinkage (crenation).
Dialysis
Principles of Dialysis
Dialysis allows solvent and small solute particles to pass through an artificial membrane, while large particles are retained. Hemodialysis is used to remove waste from blood when kidneys fail.
