 Back
BackChemical Quantities and Reactions: Study Guide
Study Guide - Smart Notes

Chemical Quantities and Reactions
Collection Terms and the Mole
In chemistry, collection terms are used to represent specific numbers of items, similar to everyday terms like "dozen" or "ream." The mole is a fundamental collection term in chemistry, representing Avogadro's number of particles.
Collection Term: Specifies a set number of items (e.g., 1 dozen = 12 items).
The Mole: 1 mole = particles (atoms, molecules, or formula units).
Atomic Mass: The atomic mass of an element in grams equals 1 mole of that element.
Example: 1 mole of Ne = 20.18 g = atoms Ne.
Mole Relationships: Atoms, Molecules, and Formula Units
The mole concept applies to both elements and compounds, allowing conversion between mass, moles, and particles.
For Elements: 1 mole contains atoms.
For Compounds: 1 mole contains molecules (covalent) or formula units (ionic).
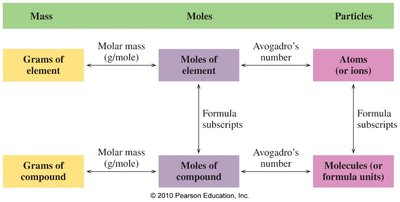
Avogadro’s Number and Conversion Factors
Avogadro’s number is used to convert between moles and particles. It can be written as an equality and as conversion factors.
Equality: 1 mole = particles
Conversion Factors: or
Example: 0.50 mol Cu × atoms/1 mol = Cu atoms
Subscripts and Moles in Chemical Formulas
Subscripts in chemical formulas indicate the ratio of atoms and moles of each element in a compound.
Example: Glucose (C6H12O6): In 1 mole, there are 6 moles of C, 12 moles of H, and 6 moles of O.
Conversion Factors: Use subscripts to relate moles of each element to moles of compound.
Molar Mass and Formula Mass
Molar mass is the mass of one mole of a substance, expressed in grams per mole (g/mol). Formula mass is the sum of atomic masses in a compound.
Molar Mass: Used as a conversion factor between grams and moles.
Example: Molar mass of CaCO3 = 100.09 g/mol
Formula Mass Calculation: Add atomic masses for each atom in the formula.

Converting Between Mass, Moles, and Particles
Conversions between mass, moles, and particles are fundamental in chemical calculations. Use molar mass and Avogadro’s number as conversion factors.
Example: 3.00 moles Al × 26.982 g Al/1 mol = 80.9 g Al
Example: 10.0 g H2S × 1 mol/34.09 g H2S = 0.293 mol H2S
Example: 10.0 g H2S × molecules/34.09 g H2S = molecules H2S
Chemical Reactions and Evidence of Change
Chemical reactions involve the transformation of substances, evidenced by color change, gas formation, precipitate formation, or heat exchange.
Reactants: Substances present before the reaction.
Products: Substances formed by the reaction.
Balanced Equations: Number and types of atoms must be equal on both sides.
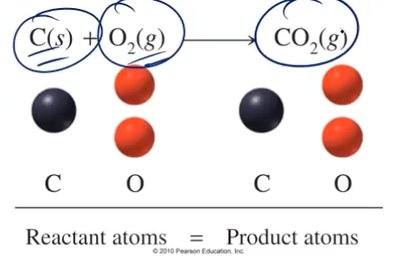
Law of Conservation of Mass
The law states that matter cannot be created or destroyed in a chemical reaction; the mass of products equals the mass of reactants.
Example: 2 moles Ag + 1 mole S = 1 mole Ag2S; 247.9 g reactants = 247.9 g products
Balancing Chemical Equations
Balancing equations ensures the conservation of mass and atoms. Only coefficients are changed, not subscripts.
Steps: Write unbalanced equation, add coefficients, reduce to smallest whole numbers, check atom balance.
Guidelines: Balance elements in one compound first, free elements last, use only coefficients, eliminate fractions.
Mole Relationships in Chemical Equations
Coefficients in balanced equations represent mole ratios, used as conversion factors in stoichiometric calculations.
Example: 4Fe(s) + 3O2(g) → 2Fe2O3(s) can be read as 4 moles Fe + 3 moles O2 → 2 moles Fe2O3
Mole-Mole Factors: , , etc.
Mass Calculations in Reactions
Stoichiometry allows calculation of product or reactant masses using mole ratios and molar masses.
Example: 2.13 × 108 g CH4 × (1 mol/16.05 g) × (1 mol CO2/1 mol CH4) × (44.01 g CO2/1 mol CO2) = 5.84 × 108 g CO2

Example: 1.80 g Na × (1 mol/22.99 g) × (2 mol NaOH/2 mol Na) × (40.00 g NaOH/1 mol NaOH) = 3.13 g NaOH
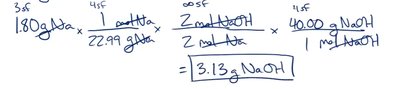
Types of Chemical Reactions
Chemical reactions are classified into several types based on the nature of reactants and products.
Combination: Two or more substances combine to form one product (A + B → AB).
Decomposition: One substance splits into two or more products (AB → A + B).
Single Replacement: One element replaces another in a compound (A + BC → AC + B).
Double Replacement: Elements in two compounds exchange places (AB + CD → AD + CB).
Combustion: Substance reacts with O2 to produce CO2, H2O, and energy.
Acid-Base: Acid reacts with base to produce water and salt.
Redox: Involves transfer of electrons between atoms.
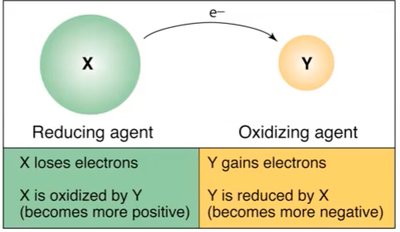
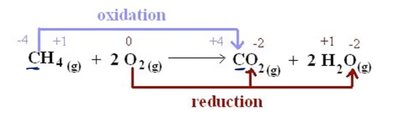
Oxidation-Reduction (Redox) Reactions
Redox reactions involve electron transfer, changing oxidation states. The reducing agent loses electrons, and the oxidizing agent gains electrons.
Reducing Agent: Loses electrons, becomes more positive.
Oxidizing Agent: Gains electrons, becomes more negative.
Example: CH4 + 2O2 → CO2 + 2H2O; CH4 is oxidized, O2 is reduced.
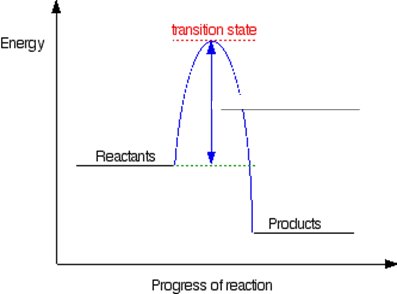
Energy in Chemical Reactions
Chemical reactions involve energy changes. The collision model explains that reactions occur when molecules collide with sufficient energy and proper orientation.
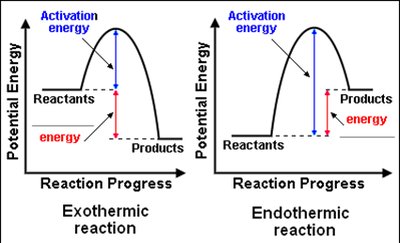
Activation Energy: Minimum energy required for a reaction to occur.
Exothermic: Releases energy; temperature increases.
Endothermic: Absorbs energy; temperature decreases.
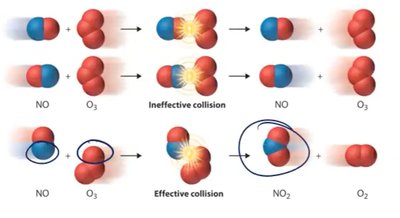
Reaction Rates and Factors Affecting Rate
The rate of a chemical reaction depends on several factors, including reactivity, accessibility, temperature, concentration, and catalysis.
Reactivity: Chemical nature of reactants affects likelihood of reaction.
Accessibility: Reactants must collide; homogeneous reactions are more favorable.
Temperature: Higher temperature increases collision frequency and energy.
Concentration: Higher concentration increases likelihood of collisions.
Catalysis: Catalysts lower activation energy and speed up reactions.
Additional info: This guide covers all major concepts from Chapter 7: Chemical Quantities and Reactions, including mole concept, stoichiometry, reaction types, energy changes, and reaction rates, with relevant examples and diagrams for clarity.
