 Back
BackChemical Quantities, The Mole, and Molar Mass: Study Notes for General Chemistry
Study Guide - Smart Notes

Chemical Quantities and Reactions
The Mole Concept
The mole is a fundamental unit in chemistry used to express amounts of a chemical substance. It allows chemists to count atoms, molecules, or ions by weighing them.
Definition: One mole (abbreviated as mol) contains exactly particles (Avogadro’s number).
Analogy: Just as a dozen means 12 items, a mole means items.
Application: 1 mole of H2O molecules = H2O molecules.

Conversion Factors: The mole can be used to convert between the number of particles and the amount in moles:
Example Calculation: How many molecules are in 3.0 moles of NaF?
Reverse Calculation: How many moles are in atoms of H2?
Key Point: Always select the conversion factor with the desired unit in the numerator.
Chemical Formulas and Formula Mass
Chemical formulas represent the types and numbers of atoms in a compound. The formula mass is the sum of the atomic masses of all atoms in the formula, measured in atomic mass units (amu).
Examples:
H2O: 2 hydrogen atoms, 1 oxygen atom
NaCl: 1 sodium atom, 1 chlorine atom
Ca(OH)2: 1 calcium ion, 2 hydroxide ions
Counting Atoms: For (NH4)2SO4:
2 ammonium ions (NH4+) = 2 N, 8 H
1 sulfate ion (SO42-) = 1 S, 4 O
Total: 2 N, 8 H, 1 S, 4 O
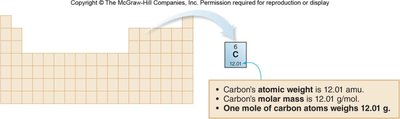
Molar Mass
The molar mass is the mass of one mole of a substance, expressed in grams per mole (g/mol). It is numerically equal to the formula mass in amu, but the units are grams per mole.
Calculation Steps:
Write the correct formula and determine the number of each atom.
Multiply the number of atoms by their atomic weights (from the periodic table).
Add the results to get the formula mass (amu) or molar mass (g/mol).
Example 1: MgSO4
1 Mg atom × 24.31 amu = 24.31 amu
1 S atom × 32.06 amu = 32.06 amu
4 O atoms × 16.00 amu = 64.00 amu
Formula mass: 120.37 amu
Molar mass: 120.37 g/mol
Example 2: H2O
2 H atoms × 1.01 amu = 2.02 amu
1 O atom × 16.00 amu = 16.00 amu
Formula mass: 18.02 amu
Molar mass: 18.02 g/mol
Example 3: CaCl2
1 Ca atom × 40.08 amu = 40.08 amu
2 Cl atoms × 35.45 amu = 70.90 amu
Formula mass: 110.98 amu
Molar mass: 110.98 g/mol
Writing Chemical Formulas for Molecular Compounds
To write the chemical formula for a molecular compound, use the prefixes in the compound’s name to determine the number of each type of atom.
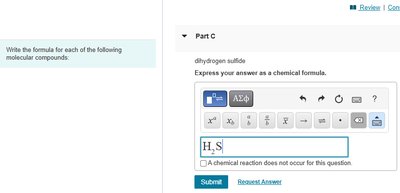
Example: Dihydrogen sulfide
"Di-" means 2 hydrogen atoms
"Sulfide" means 1 sulfur atom
Formula: H2S
Periodic Table Groups and Ionic Compounds
The group number of a metal in the periodic table often determines the charge of its ion in ionic compounds.
Group 1A: Forms +1 ions (e.g., Na+)
Group 2A: Forms +2 ions (e.g., Mg2+)
Group 3A: Forms +3 ions (e.g., Al3+)
Example: If a metal forms a compound XF3, the metal must be X3+ (Group 3A)
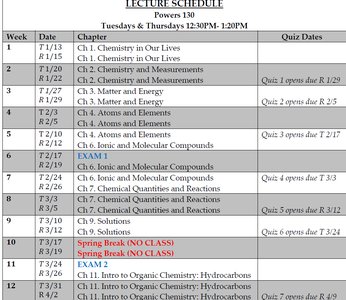
Lecture Schedule and Course Structure
The course covers foundational topics in general chemistry, including measurements, matter, atomic structure, chemical bonding, chemical reactions, solutions, acids and bases, and an introduction to organic chemistry.
Week | Date | Chapter | Quiz Dates |
|---|---|---|---|
1 | 1/13, 1/15 | Ch. 1. Chemistry in Our Lives | |
2 | 1/20, 1/22 | Ch. 2. Chemistry and Measurements | Quiz 1 opens due 1/29 |
3 | 1/27, 1/29 | Ch. 3. Matter and Energy | Quiz 2 opens due 2/5 |
4 | 2/3, 2/5 | Ch. 4. Atoms and Elements | Quiz 3 opens due 2/17 |
5 | 2/10, 2/12 | Exam 1 | |
6 | 2/17, 2/19 | Ch. 5. Nuclear Chemistry | |
7 | 2/24, 2/26 | Ch. 6. Ionic and Molecular Compounds | Quiz 4 opens due 3/3 |
8 | 3/3, 3/5 | Ch. 7. Chemical Quantities and Reactions | Quiz 5 opens due 3/12 |
9 | 3/10, 3/12 | Ch. 8. Gases | |
10 | 3/17, 3/19 | Ch. 9. Solutions | |
11 | 3/24, 3/26 | Spring Break (NO CLASS) | |
12 | 3/31, 4/2 | Ch. 11. Intro to Organic Chemistry: Hydrocarbons | Quiz 6 opens due 4/9 |
Summary Table: Molar Mass Calculations
Compound | Molar Mass (g/mol) |
|---|---|
NH4Cl | 53.50 |
C2H5OH | 46.08 |
Al2(CO3)3 | 233.99 |
Additional info: The images of flowers, colored dots, and the animal are not directly relevant to the chemical concepts discussed and are therefore excluded.
