 Back
BackChemical Reactions and Quantities: Types, Equations, and Balancing
Study Guide - Smart Notes

Chapter 7: Chemical Reactions and Quantities
Introduction to Chemical Reactions
Chemical reactions are processes in which substances (reactants) are transformed into new substances (products) with different properties and formulas. These reactions are fundamental to chemistry and are observed in everyday life, such as in cold packs used for injuries, where ammonium nitrate dissolves in water, absorbing heat and causing a temperature drop.
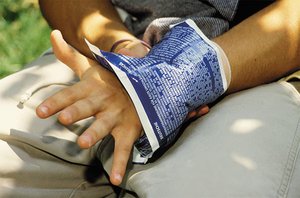
Evidence of Chemical Change
Several observable signs indicate a chemical reaction has occurred:
Change in color
Formation of a gas (bubbles)
Formation of a solid (precipitate)
Heat or light produced or absorbed

Chemical Equations
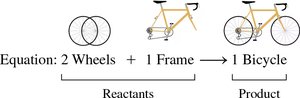
Writing and Interpreting Chemical Equations
Chemical equations represent chemical reactions using formulas for reactants and products. The reactants are written on the left, and the products on the right, separated by an arrow. Physical states are indicated in parentheses: (s) for solid, (l) for liquid, (g) for gas, and (aq) for aqueous solution.
Reactants: Substances that undergo change
Products: New substances formed
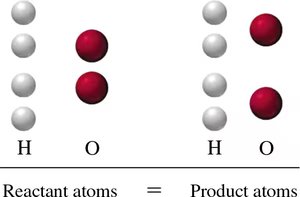
Law of Conservation of Matter
In any chemical reaction, atoms are neither created nor destroyed. This means the number of atoms of each element must be the same on both sides of the equation. This is known as the Law of Conservation of Matter.
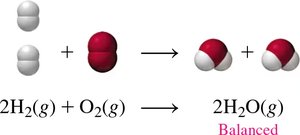
Balancing Chemical Equations
To balance a chemical equation:
Write the correct formulas for reactants and products.
Count the number of atoms of each element on both sides.
Add coefficients to balance each element.
Check that all elements are balanced using the lowest possible whole numbers.
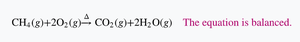
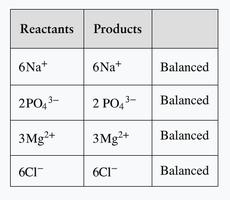
Equations with Polyatomic Ions
When the same polyatomic ion appears on both sides of the equation, it can be balanced as a group. This simplifies the balancing process.
Types of Chemical Reactions
Classification of Chemical Reactions
Chemical reactions can be classified into five main types based on the patterns of reactants and products:
Combination (Synthesis) Reactions
Decomposition Reactions
Single Replacement Reactions
Double Replacement Reactions
Combustion Reactions
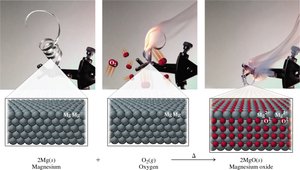
Combination Reactions
Two or more reactants combine to form a single product.

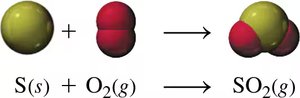
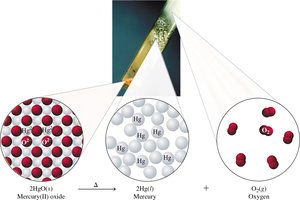
Decomposition Reactions
A single reactant splits into two or more products.

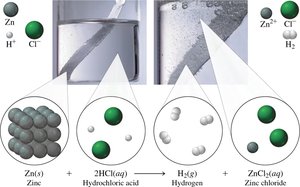
Single Replacement Reactions
One element replaces another in a compound.

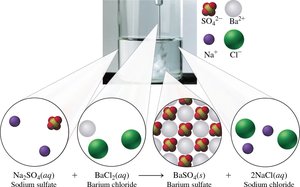
Double Replacement Reactions
The positive ions in two compounds exchange places.

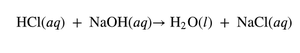
Combustion Reactions
A carbon-containing compound reacts with oxygen to produce carbon dioxide, water, and energy (heat or flame).

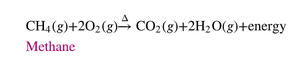
Summary Table: Types of Chemical Reactions
Reaction Type | Example |
|---|---|
Combination | |
Decomposition | |
Single Replacement | |
Double Replacement | |
Combustion |
Oxidation-Reduction (Redox) Reactions
Definition and Importance
Oxidation-reduction (redox) reactions involve the transfer of electrons from one substance to another. These reactions are essential in biological systems, batteries, and combustion processes.
Oxidation: Loss of electrons
Reduction: Gain of electrons
Example: Oxidation of Copper
When copper reacts with oxygen, copper is oxidized (loses electrons) and oxygen is reduced (gains electrons). The overall reaction is:
Cu: 0 → +2 (oxidized)
O: 0 → -2 (reduced)
Conclusion
Chemical reactions are central to chemistry, involving the transformation of substances through the breaking and forming of bonds. Understanding how to write, balance, and classify chemical equations is foundational for further study in chemistry.
