Back
BackChemical Reactions, Chemical Quantities, and Gases: Study Guide
Study Guide - Smart Notes

Chemical Reactions
Collision Theory of Reactions
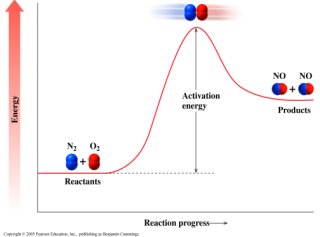
Chemical reactions occur when molecules collide with sufficient energy to break the bonds in the reactants, allowing new bonds to form in the products. The reactants must overcome an energy barrier for the reaction to proceed.
Key Point 1: Collisions must have enough energy to break old bonds and form new ones.
Key Point 2: The minimum energy required is called activation energy.
Example: Formation of nitric oxide (NO) from nitrogen (N2) and oxygen (O2).

Activation Energy
Activation energy (Ea) is the minimum energy needed for a reaction to take place. Only collisions with energy equal to or greater than Ea result in product formation.
Key Point 1: Activation energy is represented as the height of the energy barrier in a reaction progress diagram.
Key Point 2: Reactants must reach this energy threshold to transform into products.
Equation:
Exothermic and Endothermic Reactions
Reactions can be classified based on energy changes:
Exothermic Reaction: Releases heat; energy of products is less than reactants; heat is a product.
Endothermic Reaction: Absorbs heat; energy of products is greater than reactants; heat is a reactant.
Equation (Exothermic):
Equation (Endothermic):
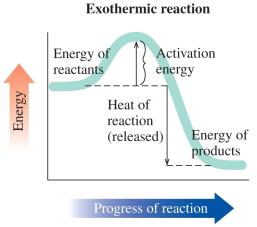
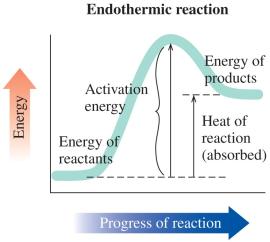
Energy Diagrams and Calculations
Energy diagrams visually represent the activation energy and heat of reaction. The difference between the energy of reactants and products determines whether the reaction is exothermic or endothermic.
Key Point 1: Activation energy is the energy difference from reactants to the peak.
Key Point 2: Heat of reaction is the energy difference between products and reactants.
Equation:
Chemical Equations and Balancing
Writing Chemical Equations
Chemical equations represent reactions using formulas and symbols. Reactants are written on the left, products on the right, and physical states are indicated.
Key Point 1: Symbols such as (s), (l), (g), (aq), and Δ are used to indicate states and conditions.
Key Point 2: The arrow (→) shows the direction of the reaction.

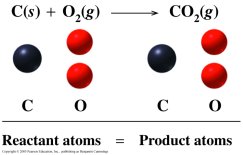
Law of Mass Conservation
The law states that atoms are neither created nor destroyed in a chemical reaction. The number of atoms of each element must be the same on both sides of the equation.
Key Point 1: Balancing equations ensures mass conservation.
Key Point 2: Coefficients are used to balance atoms, not subscripts.
Law of Constant Composition
A chemical compound always contains the same elements in the same proportions by mass. Changing subscripts alters the identity of the compound.
Key Point 1: Subscripts define the chemical identity.
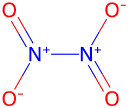
Key Point 2: Example: NO2 (nitrogen dioxide) vs. N2O4 (dinitrogen tetroxide).
Balancing Chemical Equations
Balancing equations involves adjusting coefficients to ensure equal numbers of atoms for each element on both sides, without changing subscripts.
Key Point 1: Use coefficients to balance atoms.
Key Point 2: Never change subscripts; treat polyatomic ions as units.
Example:
Types of Chemical Reactions
Combination and Combustion Reactions
Combination reactions involve two or more elements or compounds forming one product. Combustion is a special combination reaction with oxygen, producing heat and light.
Key Point 1: Combination:
Key Point 2: Combustion:
Decomposition, Single Replacement, and Double Replacement Reactions
Decomposition reactions split one substance into two or more simpler substances. Single replacement reactions involve one element replacing another in a compound. Double replacement reactions involve cations switching places between compounds.
Key Point 1: Decomposition:
Key Point 2: Single replacement:
Key Point 3: Double replacement:

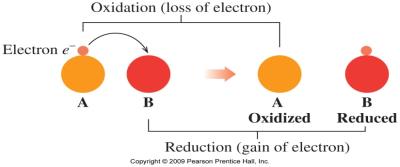
Oxidation and Reduction Reactions
Oxidation-Reduction (Redox) Reactions
Redox reactions involve the transfer of electrons between elements. Oxidation is the loss of electrons (LEO), and reduction is the gain of electrons (GER).
Key Point 1: Oxidation state helps track electron transfer.
Key Point 2: Example:
Equation:
Chemical Quantities: The Mole Concept
Collection Terms and the Mole
Collection terms represent specific numbers of items. The mole is the SI unit for amount of material, equal to units (Avogadro's number).
Key Point 1: 1 mole = atoms, molecules, or formula units.
Key Point 2: Used for counting particles in chemistry.

Mole Relationships and Conversions
Moles relate to mass, number of particles, and chemical formulas. Conversion factors allow calculation between grams, moles, and particles.
Key Point 1: Avogadro's number converts moles to particles.
Key Point 2: Subscripts in formulas indicate atom and mole ratios.
Equation:

Molar Mass and Calculations
Atomic Mass and Molar Mass
Atomic mass is the mass of an average atom in amu. Molar mass is the mass of 1 mole of an element or compound, expressed in grams per mole (g/mol).
Key Point 1: Molar mass is used to convert between grams and moles.
Key Point 2: The molar mass of a compound is the sum of the molar masses of its elements.
Equation:


Molar Mass Conversion Factors
Molar mass conversion factors relate grams and moles for the same material. They are used in stoichiometric calculations.
Key Point 1:
Key Point 2: Conversion factors: and

Stoichiometry: Mole Ratios and Calculations
Mole Ratios from Balanced Equations
Mole ratios are derived from the coefficients in balanced equations and are essential for stoichiometric calculations.
Key Point 1: Example:
Key Point 2: Mole-mole factors: ,

Gases and Gas Laws
Kinetic Molecular Theory of Gases
The kinetic molecular theory explains the behavior of gases based on the motion and energy of particles.
Key Point 1: Gas particles move rapidly and randomly, with negligible attractive forces.
Key Point 2: Gases have low density, are compressible, and have no definite shape or volume.
Properties of Gases
Gases are described by pressure (P), volume (V), temperature (T), and amount (n).
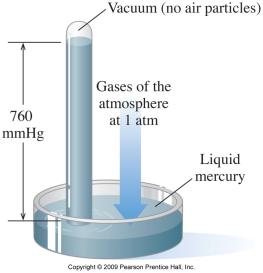
Key Point 1: Pressure is force per unit area, measured in atm, Pa, mmHg, etc.
Key Point 2: Atmospheric pressure is the pressure exerted by air from the atmosphere.

Gas Laws
Gas laws describe the relationships between P, V, T, and n.
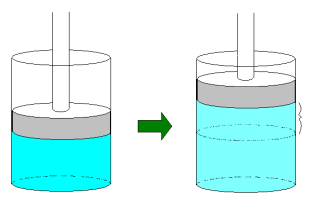
Boyle's Law: (T, n constant; pressure inversely related to volume)
Charles's Law: (P, n constant; volume directly related to temperature)
Gay-Lussac's Law: (V, n constant; pressure directly related to temperature)
Avogadro's Law: (T, P constant; volume directly related to moles)

Molar Volume and Partial Pressures
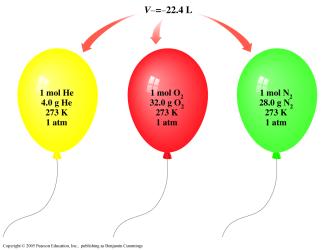
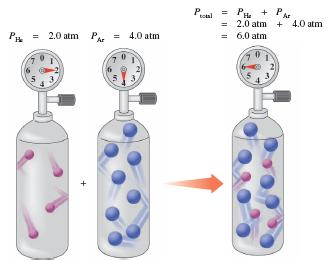
At STP (0°C, 1 atm), 1 mole of any gas occupies 22.4 L. Dalton's law states that the total pressure of a gas mixture is the sum of the partial pressures of each gas.
Key Point 1: Molar volume is used as a conversion factor for gas calculations.
Key Point 2: Partial pressure is the pressure each gas would exert alone in the container.
Equation:


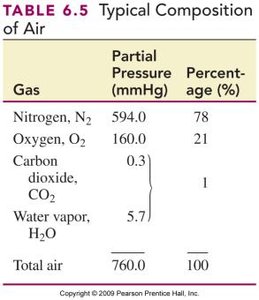
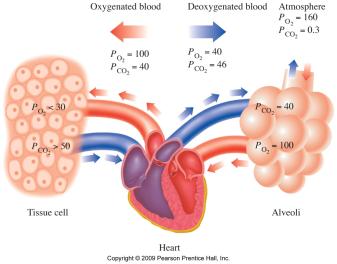
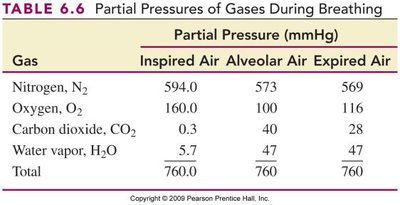
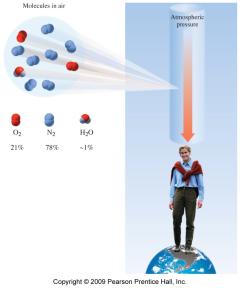
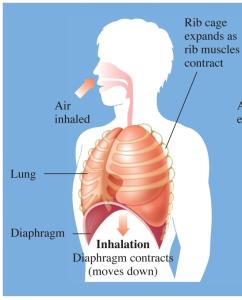
Gases in the Atmosphere and Blood
The air we breathe is a mixture of gases, mainly N2 and O2. Partial pressures are important for gas exchange in the lungs and tissues.
Key Point 1: Gas diffusion occurs from higher to lower partial pressure.
Key Point 2: Oxygen enters blood in the lungs; CO2 is released from blood.