Back
BackChemistry and Measurements: Structured Study Notes
Study Guide - Smart Notes

Chemistry and Measurements
Units of Measurement
The metric and International System of Units (SI) are the standard systems used in scientific measurement. These systems provide consistent units for volume, length, mass, temperature, and time, which are essential for accurate and reproducible scientific work.
Volume: Measured in liters (L) in the metric system and cubic meters (m3) in SI.
Length: Measured in meters (m) in both systems.
Mass: Measured in grams (g) in the metric system and kilograms (kg) in SI.
Temperature: Measured in degrees Celsius (°C) in the metric system and Kelvin (K) in SI.
Time: Measured in seconds (s) in both systems.

Example: Graduated cylinders are used to measure small volumes in milliliters (mL).

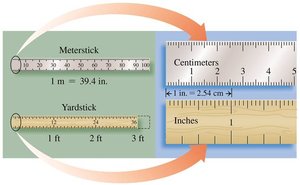
Example: Length can be measured using metersticks, yardsticks, or rulers, with conversions between centimeters and inches.
Example: Mass is measured using electronic balances, with grams and kilograms as standard units.

Example: Temperature is measured using thermometers, with Celsius and Kelvin as standard units.

Example: Time is measured using stopwatches and clocks, with seconds as the base unit.

Measured Numbers and Significant Figures
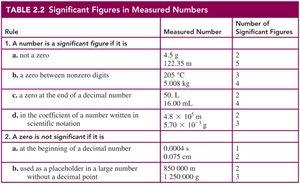
Measured numbers are obtained by measuring physical quantities and include an estimated digit. Significant figures (SFs) represent the precision of a measurement and include all nonzero digits, zeros between digits, and zeros at the end of a decimal number.
Measured Numbers: The last digit is always an estimate.
Significant Figures: Indicate the certainty of a measurement.


Table: Rules for identifying significant figures in measured numbers.
Exact Numbers
Exact numbers are obtained by counting or by definition and have an unlimited number of significant figures. They are not used to determine the number of significant figures in calculated answers.
Counted Numbers: e.g., 8 cookies, 6 eggs.
Defined Equalities: e.g., 1 kg = 1000 g, 1 qt = 4 cups.

Significant Figures in Calculations
When performing calculations, the number of significant figures in the result depends on the operation:
Multiplication/Division: The answer has the same number of SFs as the measurement with the fewest SFs.
Addition/Subtraction: The answer has the same number of decimal places as the measurement with the fewest decimal places.
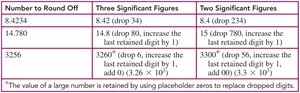
Rounding Off: If the first digit to be dropped is 5 or greater, increase the last retained digit by 1.

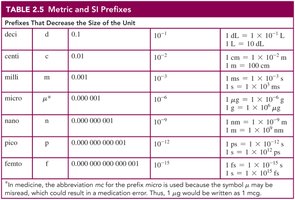
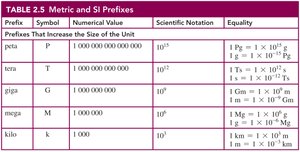
Prefixes and Equalities
Metric prefixes are used to increase or decrease the size of a unit by a factor of ten. Equalities express relationships between units and are used to write conversion factors.
Common Prefixes: milli (m), micro (μ), kilo (k), etc.
Equalities: e.g., 1 m = 100 cm, 1 L = 1000 mL.
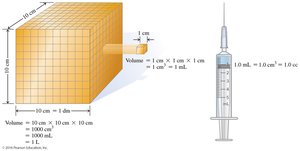
Example: 1 cm3 = 1 mL = 1 cc.

Writing Conversion Factors
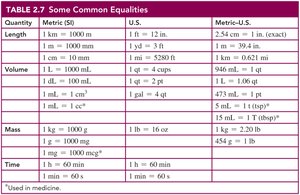
Conversion factors are fractions derived from equalities and are used to convert between units. The numbers in metric-to-metric or U.S.-to-U.S. equalities are exact, while metric-to-U.S. conversions may involve measured numbers.
Example: 1 day = 24 h can be written as or .
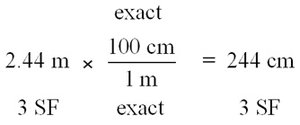
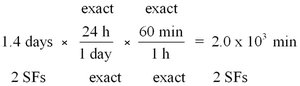
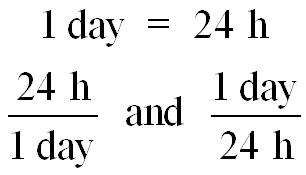
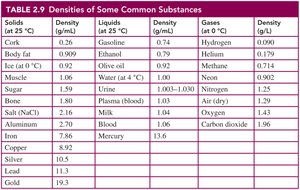
Density and Specific Gravity
Density is a physical property that compares the mass of an object to its volume. It is used to identify substances and solve problems involving mass and volume. Specific gravity is the ratio of the density of a substance to the density of water and is unitless.
Density Formula:
Specific Gravity Formula:
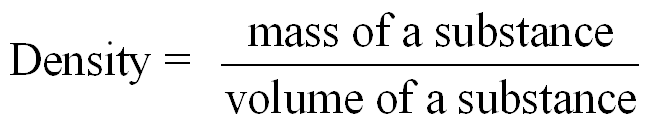
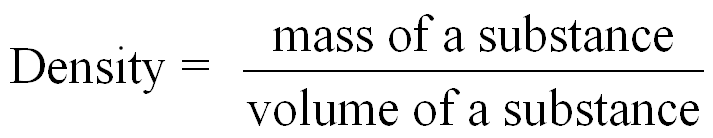
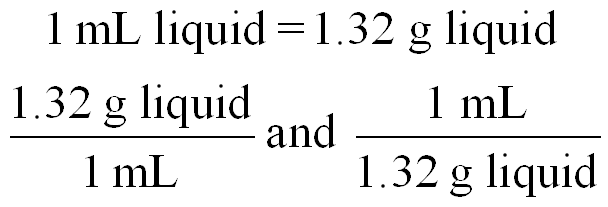
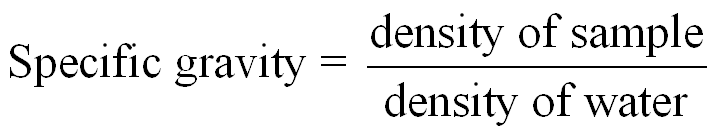
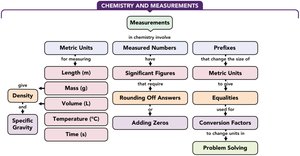
Concept Map
The concept map summarizes the relationships between measurements, metric units, significant figures, prefixes, equalities, conversion factors, density, and specific gravity in chemistry.