 Back
BackChemistry for Non-Majors I: Study Guide for Exam 2 (Chapters 6–8)
Study Guide - Smart Notes

Chapter 6: Ionic and Molecular Compounds
Octet Rule and Ions
The octet rule states that atoms tend to gain, lose, or share electrons to achieve a stable configuration of eight valence electrons, similar to noble gases.
Cations: Positively charged ions formed when metals lose electrons (e.g., Na+, Ca2+).
Anions: Negatively charged ions formed when nonmetals gain electrons (e.g., Cl-, O2-).
Ionic charges: Determined by the group number for representative elements (e.g., Group 1A forms +1, Group 2A forms +2, Group 7A forms -1).
Ionic Bonds and Compounds
Ionic bonds are formed by the transfer of electrons from metals to nonmetals, resulting in the attraction between oppositely charged ions.
Formulas: The total positive and negative charges must balance (e.g., Al3+ and Br- form AlBr3).
Naming: Name the metal first, then the nonmetal with an '-ide' ending (e.g., sodium chloride).
Metals with Variable Charges
Many transition metals can form ions with different charges. The charge is indicated with a Roman numeral in parentheses (e.g., Fe2+ is iron(II)).
Naming: For Co(NO3)2, the name is cobalt(II) nitrate.
Polyatomic Ions
Polyatomic ions are ions composed of more than one atom (e.g., NO3-, SO42-).
When writing formulas, balance the charges (e.g., Ca2+ and NO3- form Ca(NO3)2).
Name the cation first, then the polyatomic ion (e.g., ammonium sulfate).
Molecular Compounds and Covalent Bonds
Molecular (covalent) compounds are formed by the sharing of electrons between nonmetals.
Diatomic molecules: H2, N2, O2, F2, Cl2, Br2, I2.
Naming: Use prefixes to indicate the number of each atom (e.g., CO2 is carbon dioxide).
Prefixes: di- (2), tri- (3), tetra- (4), penta- (5), hexa- (6).
Lewis Structures
Lewis structures represent the arrangement of electrons in molecules. Bonds can be single, double, or triple, depending on the number of shared electron pairs.
Example: NH3 (ammonia) has three single bonds and one lone pair on nitrogen.
Electronegativity and Bond Polarity
Electronegativity is the ability of an atom to attract shared electrons. The difference in electronegativity determines bond polarity:
Nonpolar covalent: Electronegativity difference < 0.5
Polar covalent: Electronegativity difference 0.5–1.7
Ionic: Electronegativity difference > 1.7
Shapes of Molecules (VSEPR Theory)
The Valence Shell Electron Pair Repulsion (VSEPR) theory predicts the shapes of molecules based on electron pair repulsion.
Linear: 180° bond angle (e.g., CO2)
Trigonal planar: 120° (e.g., BH3)
Tetrahedral: 109.5° (e.g., CH4)
Trigonal pyramidal: (e.g., NH3)
Bent: (e.g., H2O)
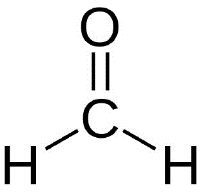
Example: The structure of formaldehyde (CH2O) is trigonal planar because the central carbon is bonded to three groups and has no lone pairs.
Intermolecular Forces
Intermolecular forces are attractions between molecules, affecting physical properties like boiling and melting points.
Dispersion forces: Weakest, present in all molecules.
Dipole-dipole attractions: Between polar molecules.
Hydrogen bonding: Strongest, occurs when H is bonded to N, O, or F.
Stronger intermolecular forces lead to higher boiling and melting points.
Chapter 7: Chemical Quantities and Reactions
The Mole and Avogadro’s Number
The mole is a counting unit in chemistry. Avogadro’s number is particles per mole.
1 mole of C atoms = C atoms
Calculations with Moles
Number of atoms/molecules from moles:
Moles from number of atoms/molecules:
Molar Mass
Molar mass is the mass of one mole of a substance (g/mol). For compounds, sum the atomic masses of all atoms in the formula.
Example: Molar mass of (NH4)2CO3 =
Mass-Mole Conversions
Mass to moles:
Moles to mass:
Balancing Chemical Equations
Equations must be balanced to obey the law of conservation of mass. Polyatomic ions can be balanced as units if unchanged.
Types of Chemical Reactions
Combination: Two or more substances form one product.
Decomposition: One substance breaks into two or more products.
Single Replacement: One element replaces another in a compound.
Double Replacement: Exchange of ions between two compounds.
Combustion: Substance reacts with O2, producing energy, CO2, and H2O.
Oxidation-Reduction (Redox) Reactions
Oxidation: Loss of electrons.
Reduction: Gain of electrons.
Example: is reduction.
Mole Relationships in Chemical Equations
Use coefficients in balanced equations to relate moles of reactants and products.
Example:
Energy in Chemical Reactions
Exothermic: Releases energy (heat).
Endothermic: Absorbs energy (heat).
Three conditions for a reaction: collision, orientation, and energy.
Rate of Reaction
Increased temperature or reactant concentration increases reaction rate.
Catalyst: Lowers activation energy, increasing reaction rate.
Chapter 8: Gases
Properties of Gases and Kinetic Molecular Theory
Gases consist of particles in constant, random motion. They have low density, are compressible, and fill their containers.
Gas Variables and Units
Pressure (P): Measured in atm, mmHg, or torr.
Volume (V): Liters (L) or milliliters (mL).
Temperature (T): Kelvin (K).
Amount (n): Moles.
Gas Laws
Boyle’s Law: (at constant T and n)
Charles’s Law: (at constant P and n)
Gay-Lussac’s Law: (at constant V and n)
Combined Gas Law:
Avogadro’s Law:
STP and Molar Volume
At STP (0°C, 1 atm), 1 mole of gas occupies 22.4 L.
Dalton’s Law of Partial Pressures
Total pressure of a gas mixture is the sum of the partial pressures of each gas:
