 Back
BackComprehensive Study Guide for GOB Chemistry Exam 2 (Ch. 5–8)
Study Guide - Smart Notes

Q4. Determine which of the statements is incorrect regarding this figure:
Background
Topic: Reaction Energy Diagrams and Activation Energy
This question tests your understanding of reaction energy diagrams, specifically how to interpret activation energy, reaction spontaneity (exergonic vs. endergonic), and reaction rates.
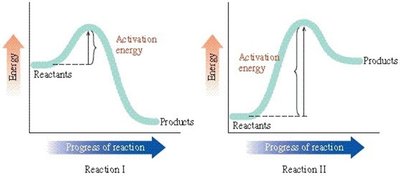
Key Terms and Concepts:
Activation Energy (Ea): The minimum energy required for a reaction to proceed.
Exergonic Reaction: A reaction where the products have lower free energy than the reactants (energy is released).
Endergonic Reaction: A reaction where the products have higher free energy than the reactants (energy is absorbed).
Reaction Rate: Influenced by the size of the activation energy barrier—the lower the barrier, the faster the reaction.
Step-by-Step Guidance
Examine the energy diagrams for Reaction I and Reaction II. Identify which reaction has a higher activation energy by comparing the height of the energy barrier from reactants to the peak.
Determine which reaction is exergonic and which is endergonic by comparing the energy levels of reactants and products in each diagram.
Recall that a lower activation energy means a reaction will generally proceed faster under the same conditions.
Review each statement in the question and use the diagrams to check if the statement matches what is shown (e.g., which reaction is faster, which is exergonic, etc.).
Try solving on your own before revealing the answer!
Final Answer: A) Reaction II occurs faster than reaction I is incorrect.
Reaction II has a higher activation energy than Reaction I, so it would proceed more slowly, not faster.
