 Back
BackGas Laws and Pressure Conversion Practice – Step-by-Step Guidance
Study Guide - Smart Notes

Q1. Convert the following barometric readings to the units requested.
Background
Topic: Pressure Unit Conversions
This question tests your ability to convert between different units of pressure, such as atmospheres (atm), millimeters of mercury (mmHg), and pounds per square inch (psi). These conversions are essential for solving gas law problems in chemistry.
Key Terms and Formulas:
1 atm = 760 mmHg
1 atm = 14.7 psi
Conversion factors are used to switch between units by multiplying or dividing as appropriate.
Step-by-Step Guidance
For each part, identify the starting unit and the unit you need to convert to.
Write the appropriate conversion factor as a fraction so that the starting unit cancels out, leaving the desired unit.
Set up the calculation for each part:
a.
b.
c.
Multiply the numbers, making sure the units cancel appropriately, and stop before calculating the final value.
Try solving on your own before revealing the answer!

Q2. The lungs of a normal adult female hold 4.2 L of air under typical atmospheric pressure (1.00 atm). If a diver holds her breath and dives to a depth of 5.0 m and the pressure increases to 1.5 atm, what is the volume of the lungs at this pressure?
Background
Topic: Boyle's Law (Pressure-Volume Relationship)
This question tests your understanding of Boyle's Law, which describes the inverse relationship between the pressure and volume of a gas at constant temperature.
Key Terms and Formulas:
Boyle's Law:
= initial pressure, = initial volume
= final pressure, = final volume (what you're solving for)
Step-by-Step Guidance
Identify the known values: , , .
Write Boyle's Law equation: .
Rearrange to solve for : .
Substitute the known values into the equation, but do not calculate the final value yet.
Try solving on your own before revealing the answer!
Q3. A pilot undergoes altitude training. He starts his training at an atmospheric pressure of 762 mmHg, where his lungs hold 6.00 L of air. He changes altitude, and his lungs expand to hold 6.25 L of air. What is the new atmospheric pressure in mmHg?
Background
Topic: Boyle's Law (Pressure-Volume Relationship)
This question also uses Boyle's Law to relate the initial and final states of a gas sample when temperature is constant.
Key Terms and Formulas:
Boyle's Law:
= initial pressure, = initial volume
= final pressure (what you're solving for), = final volume
Step-by-Step Guidance
Identify the known values: , , .
Write Boyle's Law equation: .
Rearrange to solve for : .
Substitute the known values into the equation, but do not calculate the final value yet.
Try solving on your own before revealing the answer!
Q4. If you remove a balloon with a volume of 105 mL of air from a freezer at 0°C and allow the balloon to warm to a room temperature of 25°C, what would its final volume be?
Background
Topic: Charles's Law (Volume-Temperature Relationship)
This question tests your understanding of Charles's Law, which states that the volume of a gas is directly proportional to its absolute temperature at constant pressure.
Key Terms and Formulas:
Charles's Law:
= initial volume, = initial temperature (in Kelvin)
= final volume (what you're solving for), = final temperature (in Kelvin)
Step-by-Step Guidance
Convert all temperatures to Kelvin: , .
Write Charles's Law equation: .
Rearrange to solve for : .
Substitute the known values into the equation, but do not calculate the final value yet.
Try solving on your own before revealing the answer!

Q5. A 1.20-L sample of a gas at 30°C and 1.00 atm is heated to 225°C and 1.50 atm final pressure. What is the final volume of the gas?
Background
Topic: Combined Gas Law
This question tests your ability to use the combined gas law, which relates pressure, volume, and temperature changes for a fixed amount of gas.
Key Terms and Formulas:
Combined Gas Law:
= initial pressure, = initial volume, = initial temperature (in Kelvin)
= final pressure, = final volume (what you're solving for), = final temperature (in Kelvin)
Step-by-Step Guidance
Convert all temperatures to Kelvin: , .
Write the combined gas law equation: .
Rearrange to solve for : .
Substitute the known values into the equation, but do not calculate the final value yet.
Try solving on your own before revealing the answer!
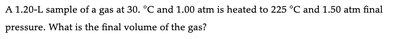
Q6. A child's balloon containing 8.5 L of helium gas at sea level is released and floats away. What will the volume of the helium be when the balloon rises to the point where the atmospheric pressure is 380 mmHg?
Background
Topic: Boyle's Law (Pressure-Volume Relationship)
This question tests your ability to apply Boyle's Law to a real-world scenario involving changes in atmospheric pressure.
Key Terms and Formulas:
Boyle's Law:
= initial pressure (sea level, 760 mmHg), = initial volume
= final pressure (380 mmHg), = final volume (what you're solving for)
Step-by-Step Guidance
Identify the known values: , , .
Write Boyle's Law equation: .
Rearrange to solve for : .
Substitute the known values into the equation, but do not calculate the final value yet.
Try solving on your own before revealing the answer!
