 Back
BackIntroduction to Organic Chemistry: Alkanes and Their Nomenclature
Study Guide - Smart Notes

Introduction to Organic Chemistry
Overview of Organic Compounds
Organic chemistry is the study of compounds primarily composed of carbon and hydrogen, often containing other elements such as oxygen, nitrogen, sulfur, and halogens. Organic compounds are the foundation of all living organisms and many synthetic materials.
Organic compounds are characterized by carbon-based structures, often forming chains or rings.
Hydrocarbons are the simplest organic compounds, containing only carbon and hydrogen.
Hydrocarbons are classified as alkanes, alkenes, alkynes, and aromatic compounds.
Alkanes
General Properties and Structure
Alkanes are saturated hydrocarbons, meaning they contain only single bonds between carbon atoms. Their general formula is , where n is the number of carbon atoms.
Each carbon atom forms four single covalent bonds.
Alkanes are nonpolar and generally unreactive except under combustion or halogenation conditions.
They are found in natural gas and petroleum.
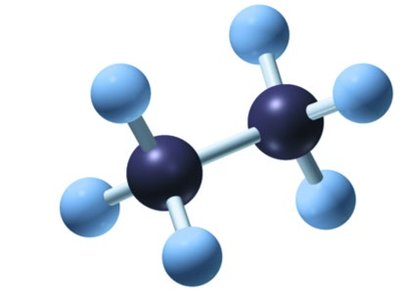
Formulas Used in Organic Chemistry
Molecular formula: Indicates the number and type of atoms (e.g., C2H6 for ethane).
Structural formula: Shows the arrangement of atoms and bonds.
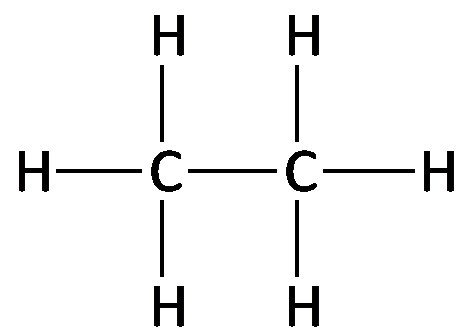
Naming Alkanes (IUPAC System)
The International Union of Pure and Applied Chemistry (IUPAC) provides systematic rules for naming alkanes. The names of alkanes end with the suffix -ane and use prefixes to indicate the number of carbon atoms.
1 carbon: Methane (CH4)
2 carbons: Ethane (C2H6)
3 carbons: Propane (C3H8)
4 carbons: Butane (C4H10)
5 carbons: Pentane (C5H12)
6 carbons: Hexane (C6H14)
7 carbons: Heptane (C7H16)
8 carbons: Octane (C8H18)
9 carbons: Nonane (C9H20)
10 carbons: Decane (C10H22)
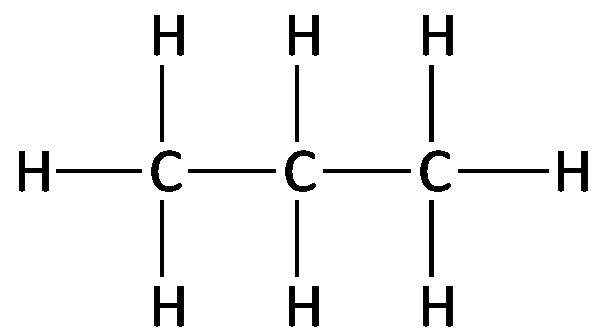
Isomerism in Alkanes
Isomers are compounds with the same molecular formula but different structural arrangements. In alkanes, constitutional (structural) isomers differ in the connectivity of their atoms.
Isomerism begins with butane (C4H10), which can exist as n-butane and isobutane (methylpropane).
Isomers have distinct physical and chemical properties.

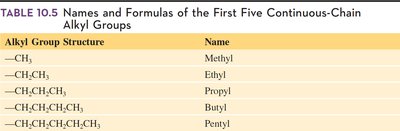
Alkyl Groups
An alkyl group is derived from an alkane by removing one hydrogen atom. Alkyl groups act as substituents on the main carbon chain and are named by replacing the -ane ending with -yl.
Alkyl Group Structure | Name |
|---|---|
—CH3 | Methyl |
—CH2CH3 | Ethyl |
—CH2CH2CH3 | Propyl |
—CH2CH2CH2CH3 | Butyl |
—CH2CH2CH2CH2CH3 | Pentyl |
Naming Branched Alkanes (IUPAC Rules)
Steps for Naming Alkanes with Substituents
When naming branched alkanes, follow these systematic steps:
Identify the longest continuous chain of carbon atoms (parent chain) and name it as the base alkane.
Number the carbon atoms in the chain starting from the end nearest a substituent (branch).
Name and locate each substituent, listing them in alphabetical order as prefixes to the parent name. Use prefixes (di-, tri-, tetra-) for multiple identical substituents, and separate numbers with commas.

Examples of Branched Alkane Structures
2-methylpropane: A three-carbon chain with a methyl group on the second carbon.
3-ethyl-2,4-dimethylhexane: A six-carbon chain with ethyl and two methyl substituents at specified positions.
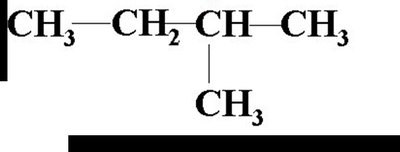
Summary Table: Steps for Naming Alkanes
Step | Description |
|---|---|
1 | Write the alkane name of the longest chain of carbon atoms. |
2 | Number the carbon atoms starting from the end nearest a substituent. |
3 | Give the location and name of each substituent (alphabetical order) as a prefix to the name of the main chain. |

Practice with Alkane Structures
Structural Isomers and Substituent Placement
Practice drawing and naming isomers by varying the position of alkyl groups on the parent chain. This reinforces the importance of numbering and alphabetical order in IUPAC nomenclature.
Draw all possible isomers for a given molecular formula.
Apply the IUPAC rules to assign correct names.
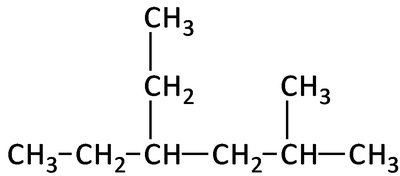
Key Terms and Concepts
Alkane: Saturated hydrocarbon with only single bonds.
Isomer: Compounds with the same molecular formula but different structures.
Alkyl group: An alkane fragment with one hydrogen removed, acts as a substituent.
IUPAC nomenclature: Systematic method for naming organic compounds.
Additional info: The content above is based on standard introductory organic chemistry curriculum and is suitable for GOB (General, Organic, and Biological) chemistry courses. The images included are directly relevant to the explanation of alkane structure, isomerism, and nomenclature.
