 Back
BackIntroduction to Organic Chemistry: Hydrocarbons and Alkanes
Study Guide - Smart Notes

Chapter 11: Introduction to Organic Chemistry
11.1 Organic Compounds
Organic compounds are a broad class of molecules that contain at least one carbon atom, typically bonded to hydrogen, and may also include oxygen, sulfur, nitrogen, or halogens. These compounds are foundational to life and are commonly found in everyday products such as fuels, medicines, plastics, and cosmetics.
Definition: Organic compounds contain carbon and hydrogen, and may include O, S, N, or halogens (Cl, Br, I).
Formula Convention: The chemical formula is written with carbon first, followed by hydrogen, then other elements.
Contrast: Inorganic compounds are typically composed of metals and nonmetals (e.g., NaCl).
Examples: Gasoline (hydrocarbons), ethanol (alcohol), and plastics (polymers).
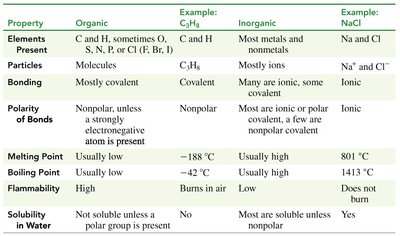
Table Purpose: The table compares the properties of organic and inorganic compounds, highlighting differences in bonding, melting/boiling points, flammability, and solubility.
11.2 Hydrocarbons
Hydrocarbons are the simplest organic compounds, consisting solely of carbon and hydrogen atoms. They can be structured as chains or rings, with each carbon atom forming four covalent bonds. Hydrocarbons are classified as either aliphatic (straight or branched chains, or rings) or aromatic (containing benzene-like rings).
Physical Properties:
Non-polar molecules; not soluble in water but soluble in nonpolar solvents.
Generally less dense than water.
Low boiling and melting points, which increase with molecular size.
Types: Alkanes, alkenes, alkynes, cycloalkanes, and aromatic hydrocarbons.
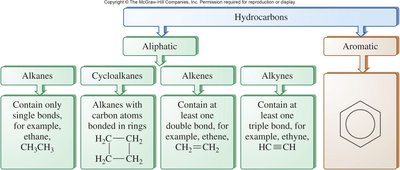
Table Purpose: This diagram classifies hydrocarbons into aliphatic (alkanes, cycloalkanes, alkenes, alkynes) and aromatic types, with examples of each.
11.3 Alkanes
Alkanes are saturated hydrocarbons, meaning they contain only single bonds between carbon atoms. Their general formula is , where n is the number of carbon atoms. Alkanes are named according to the IUPAC system, with names ending in "-ane." The first ten straight-chain alkanes have specific names and formulas.
General Formula:
Saturated: Only single C–C and C–H bonds.
Naming: Methane (1C), Ethane (2C), Propane (3C), Butane (4C), etc.
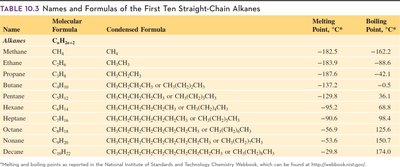
Table Purpose: This table lists the names, molecular and condensed formulas, and melting/boiling points of the first ten straight-chain alkanes.
11.4 Structural Representation of Alkanes
Alkanes can be represented in several ways to convey their structure:
Molecular Formula: Shows the number and type of atoms (e.g., C2H6 for ethane).
Structural Formula: Displays all atoms and bonds explicitly.
Condensed Formula: Groups atoms to show connectivity (e.g., CH3CH2CH3).
Line-Angle Formula: Each vertex or line end represents a carbon atom; hydrogens are implied.
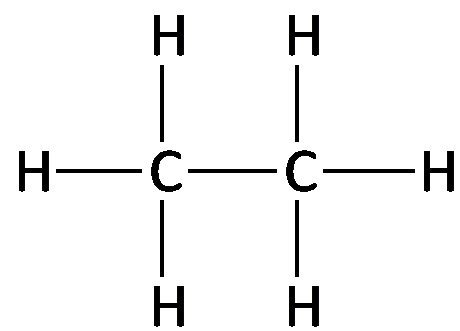
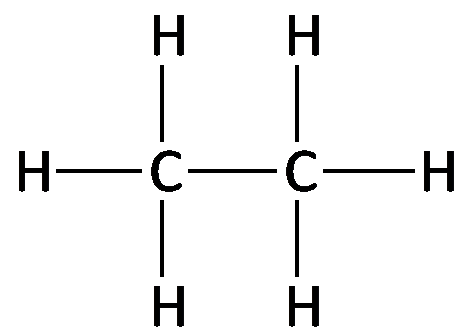
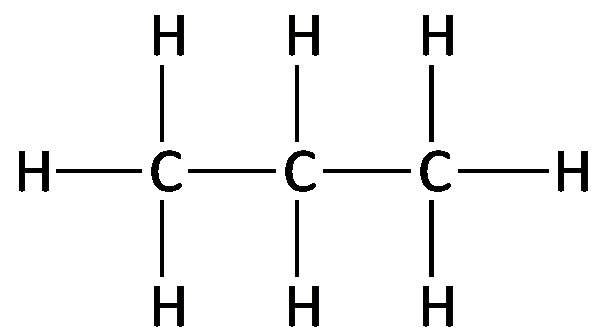
Example: The structural formula for ethane (C2H6) shows two carbon atoms each bonded to three hydrogens and to each other.
11.5 Naming Alkanes
The IUPAC system assigns names to alkanes based on the number of carbon atoms in the longest continuous chain. The first four alkanes use unique prefixes, while those with five or more carbons use Greek prefixes.
1C: Methane (CH4)
2C: Ethane (CH3CH3)
3C: Propane (CH3CH2CH3)
4C: Butane (CH3CH2CH2CH3)
5C: Pentane (CH3CH2CH2CH2CH3)
... up to Decane (10C)
11.6 Isomers and Alkyl Groups
Isomers are compounds with the same molecular formula but different structural arrangements. In alkanes with four or more carbons, branching can occur, leading to structural (constitutional) isomers. Alkyl groups are derived from alkanes by removing one hydrogen atom, and they act as substituents on the main carbon chain.
Isomers: Butane (C4H10) and methylpropane are structural isomers.
Alkyl Groups: Named by replacing the "-ane" ending with "-yl" (e.g., methyl, ethyl).
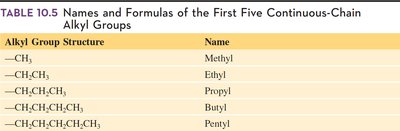
Table Purpose: This table lists the names and formulas of the first five continuous-chain alkyl groups.
11.7 Properties of Alkanes
Alkanes are generally nonpolar, insoluble in water, and have low melting and boiling points. Their physical properties change predictably with increasing molecular size.
Solubility: Insoluble in water, soluble in nonpolar solvents.
Density: Less dense than water.
Boiling/Melting Points: Increase with chain length.
Flammability: Highly flammable; burn in air to produce CO2 and H2O.
11.8 Summary Table: Properties of Organic vs. Inorganic Compounds
Property | Organic | Example: C3H8 | Inorganic | Example: NaCl |
|---|---|---|---|---|
Elements Present | C and H, sometimes O, S, N, P, or Cl (F, Br, I) | C3H8 | Most metals and nonmetals | Na and Cl |
Particles | Molecules | C3H8 | Mostly ions | Na+ and Cl- |
Bonding | Mostly covalent | Covalent | Many are ionic, some covalent | Ionic |
Polarity of Bonds | Nonpolar, unless a strongly electronegative atom is present | Nonpolar | Most are ionic or polar covalent, a few are nonpolar covalent | Ionic |
Melting Point | Usually low | -188°C | Usually high | 801°C |
Boiling Point | Usually low | -42°C | Usually high | 1413°C |
Flammability | High | Burns in air | Low | Does not burn |
Solubility in Water | Not soluble unless a polar group is present | No | Most are soluble unless nonpolar | Yes |
11.9 Summary Table: First Five Alkyl Groups
Alkyl Group Structure | Name |
|---|---|
—CH3 | Methyl |
—CH2CH3 | Ethyl |
—CH2CH2CH3 | Propyl |
—CH2CH2CH2CH3 | Butyl |
—CH2CH2CH2CH2CH3 | Pentyl |
Additional info:
Organic chemistry is essential for understanding biological molecules and industrial chemicals.
Isomerism is a key concept for understanding the diversity of organic compounds.
