 Back
BackIonic and Molecular Compounds: Structure, Bonding, and Formulas
Study Guide - Smart Notes

Ch. 6: Ionic and Molecular Compounds
Introduction to Ions and Ionic Compounds
Ions are atoms or groups of atoms that have gained or lost electrons, resulting in a net electrical charge. Ionic compounds are formed from the electrostatic attraction between positively charged cations (usually metals) and negatively charged anions (usually nonmetals). Understanding the formation, naming, and formulas of these compounds is essential in introductory chemistry.
Ions: Formation and Charges
Cations are positively charged ions formed when an atom loses electrons (typically metals).
Anions are negatively charged ions formed when an atom gains electrons (typically nonmetals).
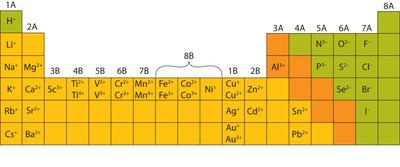
The charge of an ion is determined by its group position in the periodic table.
Transition metals can have variable charges, indicated by Roman numerals in their names (e.g., iron(II) is Fe2+, iron(III) is Fe3+).
Example: Sulfur (S) is in Group 6A, has 6 valence electrons, and gains 2 electrons to become S2−.
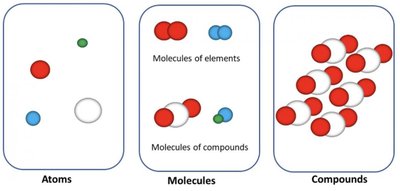
Atoms, Molecules, and Compounds
Atoms are the basic units of elements, molecules are groups of atoms bonded together, and compounds are substances composed of different elements in fixed ratios.
Atoms: Single units of elements (e.g., H, Mg, N).
Molecules: Two or more atoms chemically combined. Can be elements (e.g., H2) or compounds (e.g., H2O).
Compounds: Contain different types of atoms in fixed proportions (e.g., H2O, CO2, NaCl).
Chemical Bonds
Chemical bonds hold atoms together in molecules and compounds. The type of bond depends on the atoms involved:
Ionic bonds: Formed by the transfer of electrons from a metal to a nonmetal, resulting in the formation of ions.
Covalent bonds: Formed by the sharing of electrons between nonmetal atoms.
The octet rule states that atoms tend to gain, lose, or share electrons to achieve a stable configuration of eight valence electrons.
Chemical Formulas
A chemical formula represents the elements in a compound and the ratio of their atoms. Subscripts indicate the number of each atom present.
Example: H2O contains 2 hydrogen atoms and 1 oxygen atom.
Example: Na2SO4 contains 2 sodium, 1 sulfur, and 4 oxygen atoms.
Example: Ba(OH)2 contains 1 barium, 2 oxygen, and 2 hydrogen atoms.
Counting Atoms in a Formula
To find the total number of atoms in a formula, add the number of each type of atom.
Example: Li2SO4 has 2 Li, 1 S, and 4 O, totaling 7 atoms.
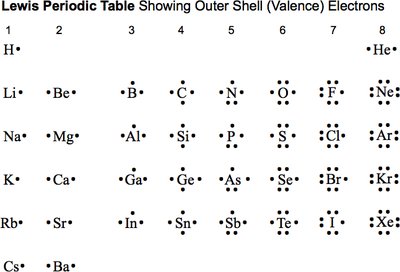
Lewis Symbols and Valence Electrons
Lewis symbols are a visual shorthand to represent the valence electrons of an atom, which are the electrons in the outermost shell and are involved in bonding. The number of valence electrons corresponds to the group number for main group elements.
Example: Nitrogen (N) is in Group 5A and has 5 valence electrons.
Types of Chemical Bonds
Ionic Bonds: Form between a cation (metal) and an anion (nonmetal) by electron transfer.
Covalent Bonds: Form between nonmetals by sharing electrons.
Writing Formulas for Ionic Compounds
To write the formula for an ionic compound:
Determine the charge of each ion.
Balance the charges so the total positive and negative charges are equal (overall charge is zero).
Write the formula with the metal (cation) first, followed by the nonmetal (anion).
Example: Magnesium (Mg2+) and chlorine (Cl−) form MgCl2.
Example: Aluminum (Al3+) and oxygen (O2−) form Al2O3.
Practice: Predicting Ionic Compound Formulas
Lithium (Li+) and oxygen (O2−): Li2O
Magnesium (Mg2+) and chlorine (Cl−): MgCl2
Sodium (Na+) and sulfur (S2−): Na2S
Calcium (Ca2+) and nitrogen (N3−): Ca3N2
Aluminum (Al3+) and oxygen (O2−): Al2O3
Summary Table: Common Ions and Their Charges
Group | Common Ion Charge | Examples |
|---|---|---|
1A | +1 | H+, Li+, Na+, K+ |
2A | +2 | Mg2+, Ca2+, Ba2+ |
3A | +3 | Al3+ |
5A | −3 | N3−, P3− |
6A | −2 | O2−, S2− |
7A | −1 | F−, Cl−, Br−, I− |
Visualizing Atoms, Molecules, and Compounds

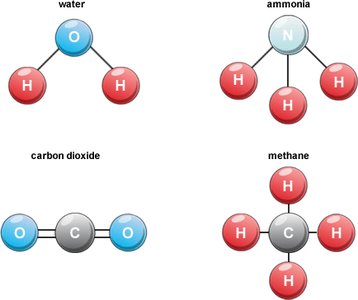
Lewis Structures for Molecules
Lewis structures use dots to represent valence electrons around atomic symbols. They help visualize bonding in molecules and predict molecular shapes.
Example: Water (H2O) has two single bonds between oxygen and hydrogen.
Example: Carbon dioxide (CO2) has two double bonds between carbon and oxygen.
Course Structure and Study Tips
Follow the lecture schedule and complete quizzes and assignments on time.
Review the study guide and practice problems before exams.
Use the periodic table and equation sheet provided during exams.
Sample Lewis Dot Structures

Sample Molecular Structures

Equations and Notation
General ionic compound formation:
Example:
Lewis structure for water:
Additional info: The provided images and tables reinforce the concepts of ion charges, Lewis structures, and molecular geometry, which are foundational for understanding ionic and molecular compounds in introductory chemistry.
