 Back
BackMatter and Energy: Temperature, Energy, and Nuclear Chemistry
Study Guide - Smart Notes

Matter and Energy
Temperature
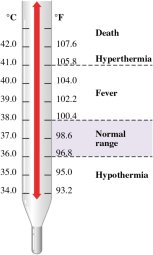
Temperature is a fundamental physical property that measures how hot or cold an object is relative to another. It is commonly measured using thermometers and is essential in understanding energy transfer and the state of matter.
Definition: Temperature quantifies the average kinetic energy of particles in a substance.
Measurement: Thermometers are used to measure temperature in degrees Celsius (°C), Fahrenheit (°F), or Kelvin (K).
Normal Body Temperature: The normal human body temperature is 37°C, which is equivalent to 310 K.
Temperature Scales: The Kelvin scale is used in scientific contexts, where .
Clinical Relevance: Body temperature ranges indicate health status, such as hypothermia, fever, and hyperthermia.
Energy
Energy is the ability to do work and is a central concept in chemistry. It exists in various forms and is involved in all chemical and physical processes.
Kinetic Energy: The energy of motion. Examples include swimming, water flowing over a dam, and exercising.
Potential Energy: Stored energy due to position or chemical composition. Examples include water at the top of a dam, compressed springs, and chemical bonds in fuels.
Heat: The energy associated with the movement of particles. The faster the particles move, the greater the heat.


Heat and Energy
Heat is a form of energy that is transferred between substances due to a temperature difference. It plays a crucial role in changes of state and chemical reactions.
Heat Transfer: When heat is added to a substance, its particles move faster, potentially causing a change of state (e.g., melting ice).
Units of Energy: Energy is measured in joules (J), kilojoules (kJ), calories (cal), and kilocalories (kcal).
Calorie Definition: One calorie is the amount of energy needed to raise the temperature of 1 g of water by 1°C.
Conversion: (exact)
Energy and Nutrition
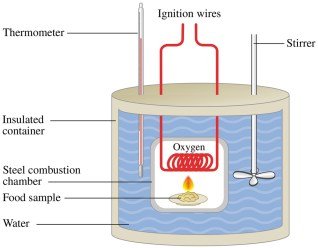
Energy values of foods are determined using calorimeters, which measure the heat released during combustion. Food labels display energy content in Calories (Cal), which are equivalent to kilocalories.
Calorimeter: A device used to measure heat transfer during the combustion of food samples.
Nutrition Labels: Energy is shown as Calories (Cal), where .
Daily Energy Requirements: Depend on gender, age, and physical activity level.
Weight Loss: Occurs when food intake is less than energy output.

Specific Heat
Specific heat is the amount of heat required to raise the temperature of 1 g of a substance by 1°C. It varies for different substances and is important in understanding heat transfer.
Formula:
Units: J/g°C or cal/g°C
Water: Has a high specific heat, which moderates temperature changes in the environment.

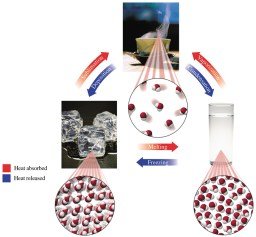
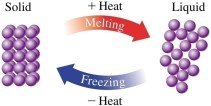
Changes of State
Matter can change from one state to another (solid, liquid, gas) through processes such as melting, freezing, evaporation, boiling, condensation, sublimation, and deposition.
Melting: Solid to liquid at the melting point.
Freezing: Liquid to solid at the freezing point.
Sublimation: Solid directly to gas.
Deposition: Gas directly to solid.
Evaporation: Liquid to gas at the surface.
Boiling: Liquid to gas throughout the liquid.
Condensation: Gas to liquid.
Heat of Vaporization: Amount of heat absorbed or released during vaporization or condensation.

Nuclear Chemistry
Natural Radioactivity
Radioactivity is the spontaneous emission of radiation from unstable atomic nuclei. It is a natural phenomenon found in certain elements and is used in medical and industrial applications.
Radioisotopes: Isotopes that emit radiation to become more stable.
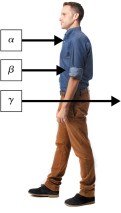
Types of Radiation: Alpha (α), beta (β), positron (β+), gamma (γ), proton (p), neutron (n).
Biological Effects: Ionizing radiation can damage cells, especially those that divide rapidly.
Radiation Protection: Different types of shielding are required for different radiation types (paper, clothing, lead, concrete).

Properties of Radiation
Radiation has distinct properties, including travel distance, tissue penetration, and required shielding. Understanding these properties is essential for safety and medical applications.
Alpha Particles: Short travel distance, blocked by paper.
Beta Particles: Moderate travel distance, blocked by heavy clothing.
Gamma Rays: Long travel distance, blocked by lead or thick concrete.
Radiation Measurement
Radiation levels are measured using devices such as Geiger counters and dosimeters. Units include curie (Ci), becquerel (Bq), rad, and rem.
Geiger Counter: Detects ionizing radiation by measuring electron flow.
Dosimeter: Worn by workers to monitor radiation exposure.
Units: Curie (Ci), Becquerel (Bq), rad, rem.


Radiation and Food
Food irradiation uses radioisotopes to extend shelf life and reduce spoilage. The FDA requires labeling of irradiated foods.
Applications: Used for fruits, vegetables, and mushrooms.
Labeling: Irradiated foods must display a specific symbol.

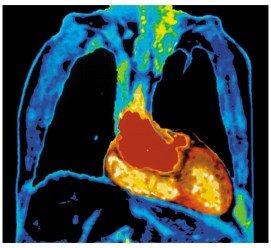
Medical Applications Using Radioactivity
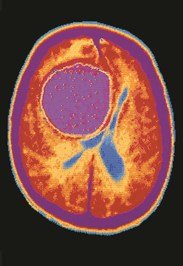
Radioisotopes are used in medical imaging and treatment, including scans, PET, CT, and MRI.
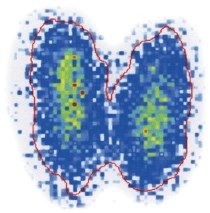
Scans: Radioisotopes such as I-131 are used to image organs like the thyroid.
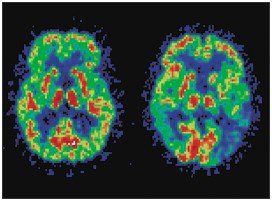
PET Scans: Show metabolic activity in the brain and can detect diseases such as Alzheimer's.
CT Scans: Use X-rays to image organs and detect abnormalities.
MRI: Uses magnetic fields and radio waves to image soft tissues without nuclear radiation.
Tables
Energy Requirements for Adults
Energy requirements vary by gender, age, and activity level.
Gender | Age | Moderately Active (kcal/kJ) | Highly Active (kcal/kJ) |
|---|---|---|---|
Female | 19–30 | 2100 (8800) | 2400 (10 000) |
Female | 31–50 | 2000 (8400) | 2200 (9200) |
Male | 19–30 | 2700 (11 300) | 3000 (12 600) |
Male | 31–50 | 2500 (10 500) | 2900 (12 100) |
Energy Expended by a 70.0-kg Adult
Activity | Energy (kcal/h) | Energy (kJ/h) |
|---|---|---|
Sleeping | 60 | 250 |
Sitting | 100 | 420 |
Walking | 200 | 840 |
Swimming | 500 | 2100 |
Running | 750 | 3100 |
Specific Heats for Some Substances
Substance | cal/g°C | J/g°C |
|---|---|---|
Aluminum, Al(s) | 0.214 | 0.897 |
Copper, Cu(s) | 0.0920 | 0.385 |
Gold, Au(s) | 0.0308 | 0.129 |
Iron, Fe(s) | 0.108 | 0.452 |
Silver, Ag(s) | 0.0562 | 0.235 |
Titanium, Ti(s) | 0.125 | 0.523 |
Ammonia, NH3(g) | 0.488 | 2.04 |
Ethanol, C2H6O(l) | 0.588 | 2.46 |
Sodium chloride, NaCl(s) | 0.207 | 0.864 |
Water, H2O(l) | 1.00 | 4.184 |
Water, H2O(s) | 0.485 | 2.03 |
Properties of Radiation and Shielding Required
Property | Alpha (α) Particle | Beta (β) Particle | Gamma (γ) Ray |
|---|---|---|---|
Travel Distance in Air | 2 to 4 cm | 200 to 300 cm | 500 m |
Tissue Depth | 0.05 mm | 4 to 5 mm | 50 cm or more |
Shielding | Paper, clothing | Heavy clothing, lab coats, gloves | Lead, thick concrete |
Typical Source | Radium-226 | Carbon-14 | Technetium-99m |
Average Annual Radiation Received by a Person in the United States
Source | Dose (mSv) |
|---|---|
Ground | 0.2 |
Air, water, food | 0.3 |
Cosmic rays | 0.4 |
Wood, concrete, brick | 0.5 |
Chest X-ray | 0.2 |
Dental X-ray | 0.2 |
Mammogram | 0.4 |
Hip X-ray | 0.6 |
Lumbar spine X-ray | 0.7 |
Upper gastrointestinal tract X-ray | 2 |
Nuclear power plants | 0.001 |
Television | 0.2 |
Air travel | 0.1 |
Radon | 2* |
Additional info: Radon exposure varies widely depending on geographic location and building materials.
