 Back
BackPharmaceutical Solutions and Tonicity: Principles and Calculations
Study Guide - Smart Notes

Pharmaceutical Solutions
Introduction to Solutions
Solutions are homogeneous mixtures composed of a solute dissolved in a solvent. In pharmaceutical sciences, solutions are widely used as dosage forms for drug delivery due to their ease of administration and rapid absorption.
Solubility: The ability of a substance (solute) to dissolve in a solvent, forming a solution. Solubility is crucial for drug effectiveness and formulation.
Types of Solutions: Solutions can be classified based on the solvent used (aqueous or non-aqueous) and their application in pharmaceutical products.
Applications: Solutions are used in oral, intravenous, ophthalmic, and topical pharmaceutical products.
Key Aspects: Important factors in solution formulation include tonicity, pH, and sterility.
Manipulating Solubility
Poorly soluble drugs require strategies to enhance their solubility for effective formulation.
Methods: Use of salts, cyclodextrins, surfactants, and selection of appropriate solvents.
Formulation Components: Preservatives, buffers, and stability enhancers are added to maintain solution quality.
Tonicity and Osmosis
Basic Principles of Osmosis
Osmosis is the movement of solvent molecules across a semi-permeable membrane from an area of higher chemical potential (pure solvent) to lower chemical potential (solution). This process is fundamental in understanding tonicity in pharmaceutical solutions.
Semi-permeable Membrane: Allows passage of solvent but not solute molecules.
Osmotic Pressure: The pressure required to prevent the movement of solvent across the membrane.
Osmolarity and Osmolality
Osmotic effects are quantified using osmolarity and osmolality, which are colligative properties (depend on the number of particles, not their identity).
Osmolarity: Number of osmotically active particles (osmol) per liter of solution.
Osmolality: Number of osmotically active particles (osmol) per kilogram of solvent.
1 Osmole: Avogadro's number of osmotically active items.
Key Values for 1 Osmolar Aqueous Solution
Osmotic pressure: 22.4 atmospheres at 0°C
Freezing point: -1.86°C
Boiling point: 100.52°C
Iso-osmotic and Iso-tonic Solutions
Iso-osmotic: Two solutions with no net movement of solvent between them, regardless of membrane or composition.
Iso-tonic: Specifically refers to biological systems (e.g., human cells). Two solutions are iso-tonic if there is no net movement of water across a biological membrane, matching the osmotic pressure of the biological system.
Importance of Iso-tonic Solutions
Maintains integrity of plasma components after injection.
Reduces pain after application.
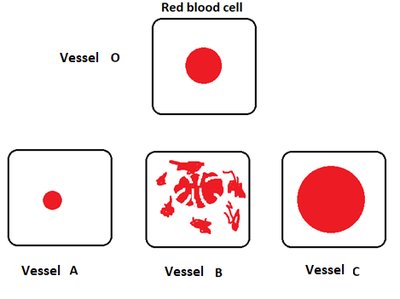
Red blood cells (RBCs) in iso-tonic medium remain normal; in hyper-tonic medium, they shrink; in hypo-tonic medium, they swell and may burst.
Calculation and Adjustment of Tonicity
Calculation Methods
Tonicity adjustment is essential for pharmaceutical solutions intended for biological systems. The goal is to match the osmolarity/osmolality of plasma (typically 325 mOsmolar).
Concentration Method: Add up the osmolarity contributions of all components and adjust to plasma values using NaCl or dextrose.
Freezing Point Depression Method: Match the freezing point depression caused by plasma and tears (-0.52°C).
NaCl Equivalent Method: Adjust to the equivalent of a 0.9% w/v NaCl solution.
Example Calculation: Concentration Method
Drug X: 250 mg / 5 mL = 50 mg/mL = 50 g/L
Number of Osmoles/L: (125 mOsmoles/L)
Adjustment required: mOsmoles/L
NaCl dissociates into 2 ions: mmoles
Amount of NaCl needed: g/L
Example Calculation: Freezing Point Depression Method
Drug X: 5% w/v
Fpd due to drug:
Adjustment required:
NaCl required: %w/v
Example Calculation: NaCl Equivalent Method
Drug X: 5% w/v
NaCl equivalent due to drug: %
Adjustment required: %
Summary Table: Tonicity Adjustment Example
Method | Drug X (g/L) | NaCl (g/L) | Water (to 1 L) |
|---|---|---|---|
Concentration | 50.00 | 5.85 | 1.00 |
Freezing Point Depression | 50.00 | 5.52 | 1.00 |
NaCl Equivalent | 50.00 | 5.50 | 1.00 |
Summary
Solutions are versatile dosage forms with advantages and disadvantages.
Formulation requires consideration of solution science, route of administration, and patient factors.
Tonicity adjustment is critical for safe and effective pharmaceutical solutions.
