 Back
BackSolutions, Acids, Bases, and Organic Compounds: Study Guide for General, Organic, and Biological Chemistry
Study Guide - Smart Notes

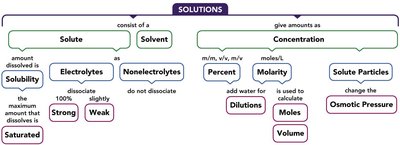
Solutions
Definition and Components
A solution is a homogeneous mixture of two or more substances. It forms when there is sufficient attraction between the solute and solvent molecules. The solvent is present in a much larger amount, while the solute is present in a smaller amount.
Solute: The substance present in lesser amount.
Solvent: The substance present in greater amount.
Example: Salt dissolved in water forms a solution.

Types of Solutes and Solvents
Solutes and solvents can be solids, liquids, or gases, resulting in various types of solutions.
Type | Example | Primary Solute | Solvent |
|---|---|---|---|
Gas in gas | Air | O2(g) | N2(g) |
Gas in liquid | Soda water | CO2(g) | H2O(l) |
Liquid in liquid | Vinegar | HC2H3O2(l) | H2O(l) |
Solid in liquid | Seawater | NaCl(s) | H2O(l) |
Solid in solid | Brass | Zn(s) | Cu(s) |
Formation of Solutions: "Like Dissolves Like"
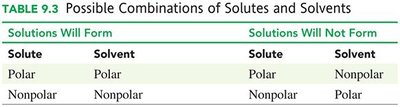
Solutions form when the solute and solvent have similar polarities. Polar solutes dissolve in polar solvents, and nonpolar solutes dissolve in nonpolar solvents.
Solutions Will Form | Solutions Will Not Form |
|---|---|
Polar solute + Polar solvent | Polar solute + Nonpolar solvent |
Nonpolar solute + Nonpolar solvent | Nonpolar solute + Polar solvent |
Electrolytes and Nonelectrolytes
Electrolytes in the Body
Electrolytes are substances that dissociate into ions in solution and conduct electricity. They are essential for proper cell and organ function. Common electrolytes include sodium, potassium, chloride, and bicarbonate.

Classification of Solutes in Aqueous Solutions
Type of Solute | In Solution | Types of Particles | Conducts Electricity | Examples |
|---|---|---|---|---|
Strong electrolyte | Dissociates completely | Only ions | Yes | NaCl, KBr, HCl, HNO3 |
Weak electrolyte | Dissociates partially | Mostly molecules, few ions | Weakly | HF, H2O, HC2H3O2 |
Nonelectrolyte | No dissociation | Only molecules | No | CH3OH, C2H5OH |

Solubility
Definition and Factors Affecting Solubility
Solubility is the maximum amount of solute that dissolves in a specific amount of solvent. It is temperature sensitive and is usually expressed as grams of solute per 100 grams of solvent.
Unsaturated solution: Contains less solute than the maximum amount.
Saturated solution: Contains the maximum amount of solute.
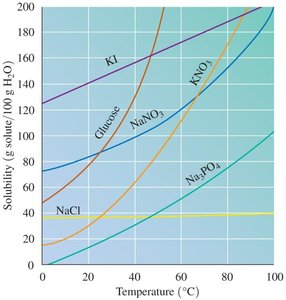
Effect of Temperature on Solubility
Solubility of most solids increases as temperature increases, while solubility of gases decreases as temperature increases.
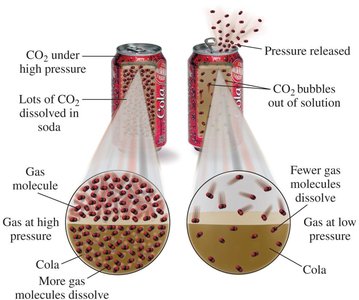
Solubility and Pressure (Henry's Law)
Henry's law states that the solubility of a gas in a liquid is directly related to the pressure of that gas above the liquid. At higher pressures, more gas molecules dissolve in the liquid.
Soluble and Insoluble Ionic Compounds
Not all ionic compounds are soluble in water. Solubility rules help predict which compounds will dissolve.
Positive Ions | Negative Ions |
|---|---|
Li+, Na+, K+, Rb+, Cs+, NH4+ | NO3-, C2H3O2- |
Cl-, Br-, I- (except with Ag+, Pb2+, Hg22+) | |
SO42- (except with Ba2+, Pb2+, Ca2+, Sr2+, Hg22+) |

Solution Concentration
Units of Concentration
The concentration of a solution is expressed in several units:
Mass percent (m/m)
Volume percent (v/v)
Mass/volume percent (m/v)
Molarity (moles solute/liters solution)
Dilution
Principles of Dilution
In a dilution, water is added, increasing the volume and decreasing the concentration, but the mass of solute remains the same. The relationship is given by:
Properties of Solutions
Isotonic Solutions
A 0.9% NaCl solution is isotonic with the solute concentration of blood cells, meaning it has the same osmotic pressure as blood.
Suspensions
Suspensions are heterogeneous mixtures with large particles that settle out and can be filtered. Examples include blood platelets and muddy water.
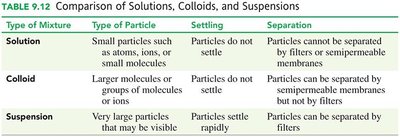
Comparison of Solutions, Colloids, and Suspensions
Type of Mixture | Type of Particle | Settling | Separation |
|---|---|---|---|
Solution | Small particles (atoms, ions, small molecules) | Do not settle | Cannot be separated by filters or membranes |
Colloid | Larger molecules or groups | Do not settle | Can be separated by membranes, not filters |
Suspension | Very large particles | Settle rapidly | Can be separated by filters |
Osmotic Pressure
Osmotic pressure is the pressure that prevents the flow of additional water into a more concentrated solution. It increases with the number of dissolved particles.
Reverse Osmosis
Reverse osmosis is a purification process where pressure greater than the osmotic pressure is applied to force water through a membrane, leaving solute molecules and ions behind.
Concept Map: Solutions
Acids and Bases
Arrhenius Acids
Arrhenius acids produce hydrogen ions (H+) when dissolved in water. They are electrolytes, have a sour taste, turn blue litmus red, and corrode some metals.
Naming Acids
Acids with H+ and a nonmetal (or CN-) are named with "hydro" and end with "ic acid" (e.g., HCl: hydrochloric acid).
Acids with H+ and a polyatomic ion: "ate" becomes "ic acid", "ite" becomes "ous acid" (e.g., HClO3: chloric acid).
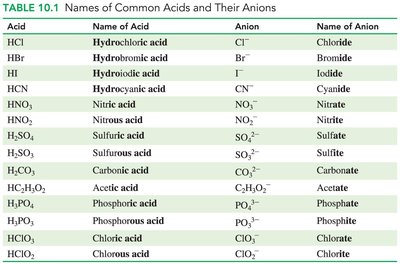
Acid | Name of Acid | Anion | Name of Anion |
|---|---|---|---|
HCl | Hydrochloric acid | Cl- | Chloride |
HBr | Hydrobromic acid | Br- | Bromide |
HI | Hydroiodic acid | I- | Iodide |
HCN | Hydrocyanic acid | CN- | Cyanide |
HNO3 | Nitric acid | NO3- | Nitrate |
H2SO4 | Sulfuric acid | SO42- | Sulfate |
HC2H3O2 | Acetic acid | C2H3O2- | Acetate |
Arrhenius Bases
Arrhenius bases produce hydroxide ions (OH-) in water. They taste bitter or chalky, feel slippery, turn litmus blue, and phenolphthalein pink.
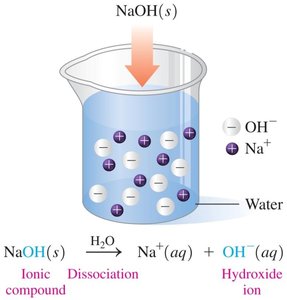
Naming Bases
Bases are named as hydroxides (e.g., NaOH: sodium hydroxide, KOH: potassium hydroxide).
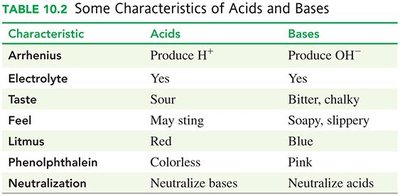
Characteristics of Acids and Bases
Characteristic | Acids | Bases |
|---|---|---|
Arrhenius | Produce H+ | Produce OH- |
Electrolyte | Yes | Yes |
Taste | Sour | Bitter, chalky |
Feel | May sting | Soapy, slippery |
Litmus | Red | Blue |
Phenolphthalein | Colorless | Pink |
Neutralization | Neutralize bases | Neutralize acids |
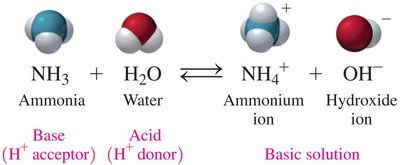
Brønsted–Lowry Acids and Bases
NH3 as a Brønsted–Lowry Base
In the reaction of ammonia and water, NH3 acts as a base (accepts H+), and H2O acts as an acid (donates H+).
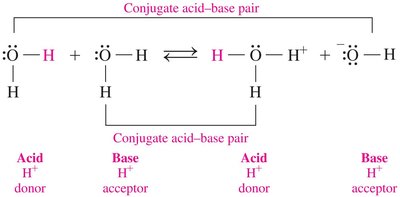
Conjugate Acid–Base Pairs
In any acid–base reaction, there are two conjugate acid–base pairs, related by the loss and gain of H+. One pair occurs in the forward direction, the other in the reverse.
Strengths of Acids and Bases
Strong and Weak Acids
Strong acids dissociate completely in water, while weak acids only partially dissociate. Most acids are weak and have strong conjugate bases.

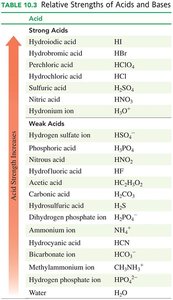
Strong and Weak Bases
Strong bases are formed from metals in Groups 1A and 2A and dissociate completely (e.g., NaOH, KOH, Ca(OH)2).
Weak bases are weak electrolytes and produce few ions in solution (e.g., NH3, Na2CO3).
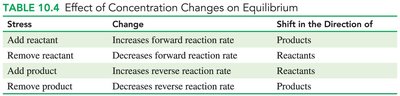
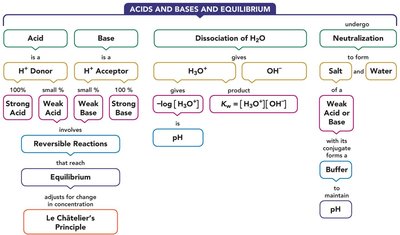
Chemical Equilibrium and Le Châtelier’s Principle
Le Châtelier’s Principle
When equilibrium is disturbed by changes in concentration, the system shifts to relieve the stress and reestablish equilibrium.
Stress | Change | Shift in Direction |
|---|---|---|
Add reactant | Increases forward rate | Products |
Remove reactant | Decreases forward rate | Reactants |
Add product | Increases reverse rate | Reactants |
Remove product | Decreases reverse rate | Products |
Ionization of Water and pH
Ionization of Water
Water is amphoteric and can act as an acid or base. H+ is transferred from one H2O molecule to another, reaching equilibrium between conjugate pairs.
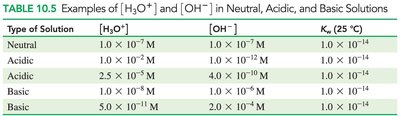
Neutral, Basic, and Acidic Solutions
The concentration of H3O+ and OH- determines whether a solution is neutral, acidic, or basic.
Type of Solution | [H3O+] | [OH-] | Kw (25°C) |
|---|---|---|---|
Neutral | 1.0 × 10-7 M | 1.0 × 10-7 M | 1.0 × 10-14 |
Acidic | 2.5 × 10-5 M | 4.0 × 10-10 M | 1.0 × 10-14 |
Basic | 5.0 × 10-11 M | 2.0 × 10-4 M | 1.0 × 10-14 |
![Comparison of [H3O+], [OH-], and pH values](https://static.studychannel.pearsonprd.tech/study_guide_files/gob/sub_images/a61c8c82_image_24.png)
pH Scale
pH is a logarithmic scale. A change of one pH unit corresponds to a tenfold change in [H3O+]. pH decreases as [H3O+] increases.
[H3O+] | pH | [OH-] |
|---|---|---|
100 | 0 | 10-14 |
10-7 | 7 | 10-7 |
10-14 | 14 | 100 |
Reactions of Acids and Bases
Acid Reactions
Acids react with certain metals to produce a salt and hydrogen gas.
Acids react with bases to produce a salt and water (neutralization).
Acids react with bicarbonate and carbonate ions to produce carbon dioxide gas.
Neutralization
In a neutralization reaction, an acid reacts with a base to produce a salt and water. The salt is formed from the anion of the acid and the cation of the base.
Acid–Base Titration
Titration is a laboratory procedure used to determine the molarity of an acid by neutralizing it with a base and using an indicator to identify the endpoint.
Buffers
Buffer Solutions
Buffers maintain pH by resisting changes when acid or base is added. They consist of a weak acid and its conjugate base (salt), or a weak base and its conjugate acid (salt).
Example: HC2H3O2(aq) and C2H3O2-(aq)
Organic Compounds
Definition and Elements
An organic compound is made from carbon and hydrogen atoms, and may contain other nonmetals such as oxygen, sulfur, nitrogen, phosphorus, or halogens. Organic compounds are found in many common products.
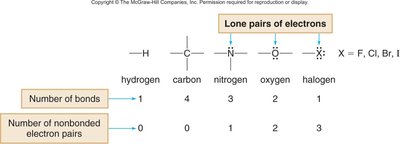
Covalent Compounds and Bonding
Covalent bonding capacity is determined by the periodic table. Carbon forms four bonds, nitrogen three, oxygen two, and halogens one.
Alkanes
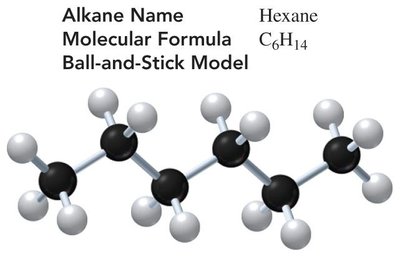
Structure and Naming
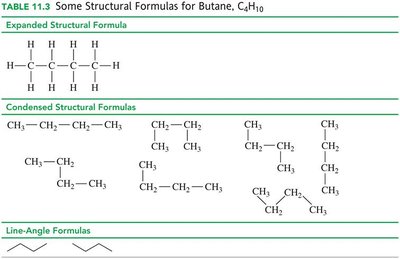
Alkanes are hydrocarbons with only single bonds. The covalent bond between carbon atoms is very strong, allowing for many possible compounds. Alkanes can be represented by molecular, expanded, condensed, and line-angle formulas.
Alkanes with Substituents
When an alkane has four or more carbon atoms, side groups (branches or substituents) can be attached. The IUPAC naming system is used for these compounds.
Structural Isomers
Structural isomers have the same molecular formula but different arrangements of atoms. For example, butane (C4H10) has a straight chain and a branched chain isomer.
Naming Cycloalkanes with Substituents
When one substituent is attached to a cycloalkane, its name is placed in front of the cycloalkane name. No number is needed for a single alkyl group or halogen atom.
Drawing Formulas for Alkanes
The IUPAC name provides all information needed to draw the condensed structural formula for an alkane.
Properties of Alkanes
Alkanes have physical properties such as solubility and density that determine their uses. Solid alkanes are used as waxy coatings on fruits and vegetables to retain moisture and inhibit mold.
Alkenes and Alkynes
Structure and Naming
Alkenes contain double bonds, and alkynes contain triple bonds. They are unsaturated hydrocarbons and react with hydrogen gas to become alkanes. The IUPAC names for alkenes and alkynes are similar to alkanes, with "ene" and "yne" endings.
Cis–Trans Isomers of Alkenes
Cis isomers have alkyl groups on the same side of the double bond, while trans isomers have them on opposite sides.
Addition Reactions
Alkenes and alkynes undergo addition reactions, where double and triple bonds are broken to add new atoms.
Aromatic Compounds
Benzene and Derivatives
Benzene is an aromatic compound with a ring of six carbon atoms and alternating double bonds. Aromatic compounds with a benzene ring and a single substituent are named as benzene derivatives.
Naming Aromatic Compounds with Substituents
When toluene, phenol, or aniline has substituents, the carbon attached to the functional group is numbered as carbon 1, and other substituents are named alphabetically.
Concept Maps
Additional info: Academic context and explanations have been expanded for clarity and completeness. All included images are directly relevant to the adjacent content and reinforce key concepts.
