 Back
BackStep-by-Step Guidance for Alkene to Alkyl Halide Reactions
Study Guide - Smart Notes

Q4. What alkene is needed as a starting material to prepare each of the following alkyl halides or dihalides? In each case, also indicate what reagent is needed to react with the alkene.
Background
Topic: Electrophilic Addition Reactions of Alkenes
This question tests your understanding of how alkenes react with hydrogen halides (like HBr or HCl) to form alkyl halides. You need to identify the correct alkene starting material and the reagent required for the transformation.
Key Terms and Formulas
Alkene: A hydrocarbon with a carbon-carbon double bond (C=C).
Alkyl halide: An organic compound containing a halogen atom (Cl, Br, I) bonded to an sp3 carbon.
Electrophilic Addition: The reaction where an electrophile (like H+ from HBr or HCl) adds to the double bond of an alkene, followed by nucleophilic attack by the halide ion.
Markovnikov's Rule: In the addition of HX to an unsymmetrical alkene, the hydrogen attaches to the carbon with more hydrogens, and the halide attaches to the carbon with fewer hydrogens.
Step-by-Step Guidance
For each product, look at the structure of the alkyl halide and identify where the halogen (Br or Cl) is attached. This will help you determine the position of the double bond in the starting alkene.
Work backwards: Remove the halogen atom and replace it with a double bond between the carbon that was bonded to the halogen and an adjacent carbon. This gives you the structure of the alkene starting material.
Identify the reagent needed for the reaction. For example, if the product is an alkyl bromide, the reagent is usually HBr. If the product is an alkyl chloride, the reagent is HCl.
Apply Markovnikov's rule to predict the major product if the alkene is unsymmetrical. The halogen will add to the more substituted carbon (the one with fewer hydrogens).
Draw the reaction showing the alkene, the reagent (HBr or HCl), and the product (alkyl halide).
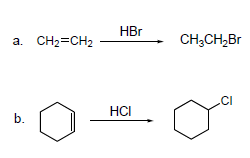
Try solving on your own before revealing the answer!
Final Answer:
a. The starting alkene is ethene (CH2=CH2), and the reagent is HBr. The product is bromoethane (CH3CH2Br).
b. The starting alkene is cyclohexene, and the reagent is HCl. The product is chlorocyclohexane.
In both cases, the halogen adds across the double bond according to Markovnikov's rule.
