 Back
BackUnit 2 Study Guide: Physical and Chemical Changes, States of Matter, Ions, Ionic and Molecular Compounds
Study Guide - Smart Notes

Structure of the Atom and Valence Electrons
Atomic Structure and Electron Cloud
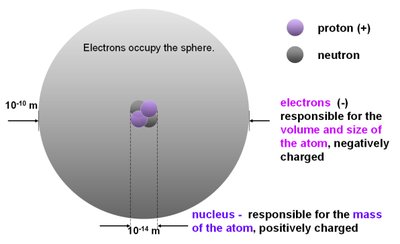
The atom consists of a central nucleus containing protons and neutrons, surrounded by an electron cloud. The arrangement of electrons, especially the valence electrons, determines the chemical properties of an element.
Protons: Positively charged particles in the nucleus.
Neutrons: Neutral particles in the nucleus.
Electrons: Negatively charged particles occupying energy levels around the nucleus.
Valence Electrons: Electrons in the highest energy level, crucial for chemical reactions.
Energy Levels are numbered (n = 1, 2, 3, ...), with higher numbers indicating greater energy and distance from the nucleus. Valence electrons are found in the outermost energy level.
Lewis Symbols
Lewis (electron-dot) symbols represent valence electrons as dots around the element symbol, with no more than two dots per side.
Example: Calcium (Ca) has two dots, Carbon (C) has four, Sulfur (S) has six.
Ions and Ionic Compounds
Formation of Ions
Atoms gain or lose electrons to achieve a stable electron configuration, often an octet (eight valence electrons).
Cations: Formed by metals losing electrons; positive charge.
Anions: Formed by nonmetals gaining electrons; negative charge.
Octet Rule: Atoms tend to gain, lose, or share electrons to reach eight in their valence shell (except H, Li, Be, which follow the duet rule).
Ion Charges and Naming
Group 1A metals: +1 charge
Group 2A metals: +2 charge
Group 3A metals: +3 charge
Group 5A, 6A, 7A nonmetals: -3, -2, -1 charges respectively
Ion charges are shown as superscripts (e.g., Ca2+, O2−)
Ionic Compounds
Ionic compounds consist of cations and anions held together by ionic bonds. The formula shows the cation first, followed by the anion, with subscripts indicating the number of each ion to ensure a net charge of zero.
High melting and boiling points
Solid at room temperature



Writing and Naming Ionic Compounds
Formula: Cation symbol (with charge) + Anion symbol (with charge)
Balance charges by adjusting subscripts
Names: Cation name (with Roman numeral if variable charge) + Anion name (root + "-ide" for monatomic anions)
Formula | Cation | Anion | Compound Name |
|---|---|---|---|
KCl | K+ | Cl− | potassium chloride |
CaI2 | Ca2+ | I− | calcium iodide |
Al2O3 | Al3+ | O2− | aluminum oxide |
Fe2S3 | Fe3+ | S2− | iron(III) sulfide |
CuO | Cu2+ | O2− | copper(II) oxide |
Periodic Table and Predictable Charges
Some metal cations have predictable charges based on their group number. Others require Roman numerals to indicate their charge.
States of Matter and Changes of State
Solids, Liquids, and Gases
Matter exists in three primary states: solid, liquid, and gas. Each state has distinct properties regarding shape, volume, and particle arrangement.
Solids: Definite shape and volume; particles in fixed arrangement, moving slowly.
Liquids: Indefinite shape, definite volume; particles close together but mobile.
Gases: Indefinite shape and volume; particles far apart, moving rapidly.




Temperature and Temperature Scales
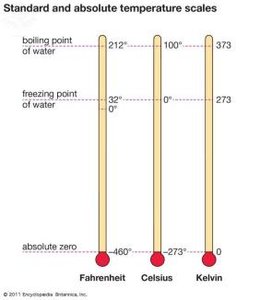
Temperature measures the average kinetic energy of particles and determines the direction of heat flow. Common scales are Celsius (°C), Fahrenheit (°F), and Kelvin (K).
Celsius: Water freezes at 0°C, boils at 100°C
Fahrenheit: Water freezes at 32°F, boils at 212°F
Kelvin: Absolute zero is 0 K; water freezes at 273 K, boils at 373 K

Temperature Conversion Formulas
To convert Celsius to Fahrenheit:
To convert Fahrenheit to Celsius:
To convert Celsius to Kelvin:
Physical Changes and Phase Changes
Physical changes alter the state, shape, or size of a substance without changing its identity. Phase changes are physical changes that require or release heat.
Endothermic: Melting, vaporization, sublimation (heat absorbed)
Exothermic: Freezing, condensation, deposition (heat released)






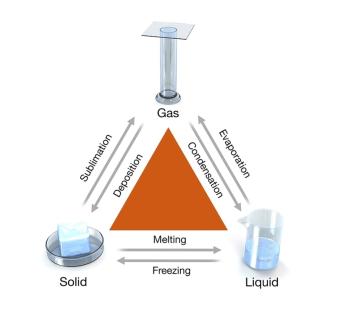
Heat of Fusion and Heat of Vaporization
Heat of Fusion: Amount of heat required to melt 1 g of a solid or released when 1 g of liquid freezes.
For water: or
Heat of Vaporization: Amount of heat required to vaporize 1 g of liquid or released when 1 g of gas condenses.
For water: or
Chemical Changes and Reactions
Chemical Changes
Chemical changes involve the formation of new substances with different compositions and properties. Chemical reactions break old bonds and form new ones, rearranging atoms.
Heat may be released (exothermic) or required (endothermic).


Examples of Physical vs. Chemical Changes
Physical Changes | Chemical Changes |
|---|---|
Boiling water | Splitting water into hydrogen and oxygen |
Tearing paper | Burning paper |
Melting aluminum | Reacting aluminum with hydrochloric acid |
Dissolving sugar in water | Fermenting sugar-water with yeast |
Cutting bread | Digesting bread |
Polyatomic Ions
Definition and Examples
A polyatomic ion is a group of atoms bonded together with an overall ionic charge. Most are anions, except ammonium (NH4+).
Examples: Nitrate (NO3−), Sulfate (SO42−), Phosphate (PO43−), Hydroxide (OH−), Ammonium (NH4+)

Writing Formulas with Polyatomic Ions
Cation is written first, followed by the polyatomic ion.
Parentheses are used if more than one polyatomic ion is present (e.g., Mg(NO3)2).
Naming Compounds with Polyatomic Ions
Name the cation first, then the polyatomic anion.
Examples: NaNO3 (sodium nitrate), K2SO4 (potassium sulfate), Fe(HCO3)3 (iron(III) bicarbonate)
Covalent (Molecular) Compounds
Covalent Bonds and Molecules
Covalent bonds form between two nonmetal atoms by sharing valence electrons to complete octets (or duets for hydrogen).
Examples: H2, N2, CO2, SO42−
Electron pairs not shared are called lone pairs.
Naming Covalent Compounds
First nonmetal is named fully; second uses root + "-ide"
Prefixes indicate the number of atoms: mono-, di-, tri-, tetra-, penta-, hexa-, hepta-, octa-, nona-, deca-
Example: SO3 is sulfur trioxide; P4S3 is tetraphosphorus trisulfide

Writing Covalent Compound Formulas
List element symbols in order given by the name
Use subscripts according to prefixes
Do not reduce subscripts
Identifying Ionic vs. Covalent Compounds
Ionic: Formula starts with a metal or NH4+
Covalent: Formula starts with a nonmetal
Electronegativity and Types of Bonds
Electronegativity Trends
Electronegativity measures an atom's attraction for shared electrons. It increases across a period and decreases down a group. Nonmetals have high electronegativity; metals have low.


Bond Types Based on Electronegativity Difference
Nonpolar Covalent: Difference ≤ 0.4; equal sharing
Polar Covalent: Difference > 0.4 and ≤ 1.8; unequal sharing, partial charges (δ+ and δ−)
Ionic: Difference > 1.8; electron transfer, full charges
Atoms | Electronegativity Difference | Type of Bond |
|---|---|---|
N-N | 0.00 | Nonpolar covalent |
C-H | 0.35 | Nonpolar covalent |
N-F | 0.94 | Polar covalent |
Cl-K | 2.34 | Ionic |
Partial Charges: In polar covalent bonds, the more electronegative atom has a partial negative charge (δ−), and the less electronegative atom has a partial positive charge (δ+).
