 Back
BackVisual Representations of Matter: Pure Substances, Mixtures, and Elements
Study Guide - Smart Notes

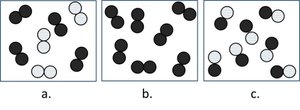
Q13. Which picture best represents a compound?
Background
Topic: Classification of Matter
This question tests your ability to distinguish between pure substances (elements and compounds) and mixtures based on visual representations.
Key Terms:
Compound: A substance composed of two or more different types of atoms chemically bonded together.
Element: A substance composed of only one type of atom.
Mixture: A combination of two or more substances that are not chemically bonded.
Step-by-Step Guidance
Examine each picture for the presence of different types of atoms (often shown as different colors or shades).
Look for pairs or groups of atoms that are consistently bonded together, indicating a compound.
Compare the arrangement: a compound should show molecules made of two or more different atoms bonded together, not just a mixture of separate atoms or molecules.
Try solving on your own before revealing the answer!
Final Answer: Picture c
Picture c shows molecules made up of two different types of atoms bonded together, which is characteristic of a compound.
Q14. Which picture best represents a heterogeneous mixture?
Background
Topic: Classification of Matter
This question tests your understanding of mixtures, specifically the difference between homogeneous and heterogeneous mixtures.
Key Terms:
Heterogeneous Mixture: A mixture in which the components are not uniformly distributed and can be visually distinguished.
Homogeneous Mixture: A mixture in which the components are uniformly distributed.
Step-by-Step Guidance
Look for a picture where different types of particles are not evenly mixed and can be seen as separate regions.
Compare the distribution of particles in each box: a heterogeneous mixture will have visible differences in composition throughout.

Try solving on your own before revealing the answer!
Final Answer: Picture a
Picture a shows visibly different types of particles in separate regions, which is characteristic of a heterogeneous mixture.
Q15. Which picture best represents a pure element?
Background
Topic: Classification of Matter
This question tests your ability to identify a pure element based on visual representation.
Key Terms:
Pure Element: A substance made up of only one type of atom.
Step-by-Step Guidance
Look for a picture where all particles are identical and consist of only one type of atom.
Check for uniformity: a pure element will not have different types of atoms or molecules.

Try solving on your own before revealing the answer!
Final Answer: Picture b
Picture b shows only one type of atom throughout, which is characteristic of a pure element.
