 Back
BackAcids and Bases: Properties, Reactions, and Quantitative Analysis
Study Guide - Smart Notes

Acids and Bases
Introduction to Acids and Bases
Acids and bases are fundamental chemical substances with distinct properties and behaviors. Their study is essential in understanding chemical reactions, solution chemistry, and biological processes.
Acids are substances that produce hydrogen ions (H+) in solution.
Bases are substances that produce hydroxide ions (OH-) in solution.
Acids and bases are classified by their strength, reactions, and definitions (Arrhenius and Brønsted–Lowry).

Properties of Acids
Physical and Chemical Properties
Acids exhibit characteristic properties that distinguish them from other substances.
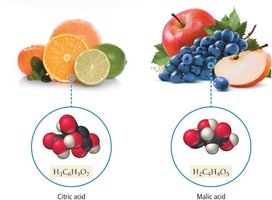
Sour taste (e.g., citric acid in lemons).
Dissolve many metals (except noble metals like gold).
Turn blue litmus paper red due to their effect on indicators.
Common Acids
Hydrochloric acid (HCl): Found in stomach acid and used in industry.
Sulfuric acid (H2SO4): Used in batteries and manufacturing.
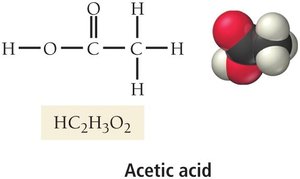
Acetic acid (HC2H3O2): Responsible for the sour taste of vinegar.
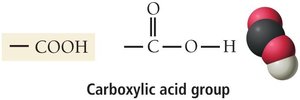
Carboxylic acids: Contain the carboxylic acid group (–COOH), found in many biological substances.

Properties of Bases
Physical and Chemical Properties
Bases are substances with distinct characteristics.
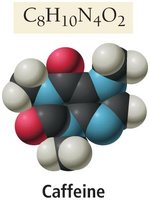
Bitter taste (e.g., caffeine in coffee).
Slippery feel (e.g., soap).
Turn red litmus paper blue.
Common Bases
Sodium hydroxide (NaOH): Used in cleaning products and manufacturing.
Potassium hydroxide (KOH): Used in soap production.
Sodium bicarbonate (NaHCO3): Found in baking soda and antacids.
Alkaloids: Organic bases found in plants, often poisonous (e.g., coniine).
Definitions of Acids and Bases
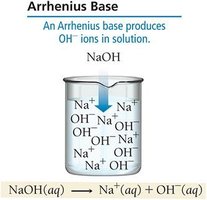
Arrhenius Definition
The Arrhenius definition is based on ion production in aqueous solution.
Acid: Produces H+ ions in solution.
Base: Produces OH- ions in solution.

Brønsted–Lowry Definition
The Brønsted–Lowry definition expands the concept to proton transfer.
Acid: Proton donor.
Base: Proton acceptor.
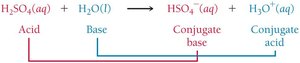
Acid–base reactions involve the transfer of a proton (H+).
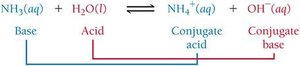
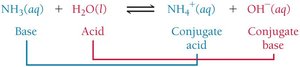
Conjugate Acid-Base Pairs
Concept and Examples
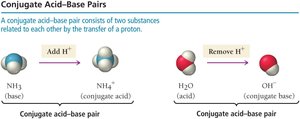
A conjugate acid-base pair consists of two substances related by the transfer of a proton.
When a base accepts a proton, it becomes a conjugate acid.
When an acid donates a proton, it becomes a conjugate base.
Reactions of Acids and Bases
Neutralization Reactions
Neutralization occurs when an acid reacts with a base to form water and a salt.
General equation:
Net ionic equation:


Acid-Metal Reactions
Acids react with metals to produce hydrogen gas and a salt.
General equation:

Acid-Metal Oxide Reactions
Acids react with metal oxides to produce water and a salt.
Example:
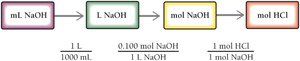
Acid–Base Titration
Quantitative Analysis of Acids and Bases
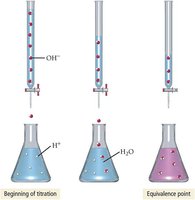
Titration is a laboratory technique used to determine the concentration of an acid or base.
A solution of known concentration is added to a solution of unknown concentration until the reaction reaches the equivalence point.
The equivalence point is signaled by an indicator (e.g., phenolphthalein).
Stoichiometry is used to calculate the unknown concentration.


Strong and Weak Acids and Bases
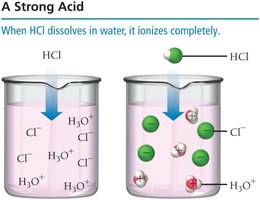
Strong Acids
A strong acid completely ionizes in solution, producing only ions.
Example:
Strong acids are strong electrolytes and conduct electricity well.

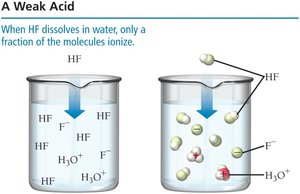
Weak Acids
A weak acid only partially ionizes in solution, leaving many intact molecules.
Example:
Weak acids are weak electrolytes and conduct electricity poorly.

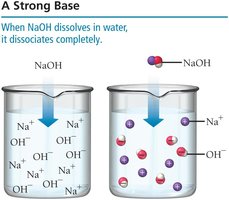
Strong Bases
A strong base completely dissociates in solution.
Example:
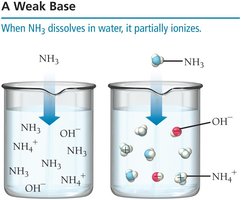
Weak Bases
A weak base only partially ionizes, often by accepting a proton from water.
Example:
Self-Ionization of Water and the Ion Product Constant
Self-Ionization of Water
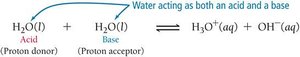
Water can act as both an acid and a base, undergoing self-ionization.
Equation:
Produces equal concentrations of H3O+ and OH- in pure water.
Ion Product Constant for Water ()
At 25°C:
In a neutral solution: M






The pH Scale
Definition and Calculation
The pH scale expresses the acidity or basicity of a solution based on hydrogen ion concentration.
pH < 7: Acidic solution
pH > 7: Basic solution
pH = 7: Neutral solution
pH is calculated as:
Logarithmic Nature of pH
A decrease of 1 unit in pH corresponds to a tenfold increase in H3O+ concentration.
Buffers
Buffer Solutions and Their Function
Buffers are solutions that resist changes in pH upon addition of acid or base.
Contain significant amounts of a weak acid and its conjugate base.
The weak acid neutralizes added base; the conjugate base neutralizes added acid.
Human blood is an important biological buffer.
Chemistry and Health
Alkaloids
Alkaloids are organic bases found in plants, often with medicinal or toxic properties (e.g., morphine, amphetamine, caffeine, nicotine).
Antifreeze Poisoning
Ethylene glycol in antifreeze is metabolized to glycolic acid, which can overwhelm blood buffers and cause dangerous drops in blood pH.
Summary Table: Strong Acids and Bases
Strong Acids | Strong Bases |
|---|---|
Hydrochloric acid (HCl) | Sodium hydroxide (NaOH) |
Hydrobromic acid (HBr) | Potassium hydroxide (KOH) |
Hydroiodic acid (HI) | Calcium hydroxide (Ca(OH)2) |
Nitric acid (HNO3) | Barium hydroxide (Ba(OH)2) |
Perchloric acid (HClO4) | Strontium hydroxide (Sr(OH)2) |
Sulfuric acid (H2SO4) | — |
Additional info: Table entries inferred from standard textbook content.
Key Equations
Learning Objectives
Identify common acids and bases and describe their properties.
Distinguish between Arrhenius and Brønsted–Lowry definitions.
Write equations for acid-base reactions, including neutralization and reactions with metals and metal oxides.
Use titration to determine unknown concentrations.
Identify strong and weak acids and bases.
Calculate pH, [H3O+], and [OH-].
Describe how buffers resist pH change.
