 Back
BackAcids and Bases: Properties, Theories, and Applications
Study Guide - Smart Notes

Acids and Bases
Experimental Definitions
Acids and bases are fundamental chemical substances with distinct properties and behaviors. Their identification in laboratory and everyday contexts relies on observable characteristics.
Acids:
Taste sour.
Turn litmus paper red.
React with active metals to release hydrogen gas.
React with bases to form water and a salt.
Bases:
Taste bitter.
Turn litmus paper blue.
Feel slippery to the touch.
React with acids to form water and a salt.

Arrhenius Theory
The Arrhenius theory provides a classical definition of acids and bases based on their behavior in aqueous solutions.
Acid: A molecular substance that ionizes in water to produce hydrogen ions ().
Base: A substance that produces hydroxide ions () in water.
Common Acids and Bases
Name | Formula | Acid/Base Strength | Common Uses/Notes |
|---|---|---|---|
Sulfuric acid | H2SO4 | Strong | Battery acid, fertilizer manufacturing |
Hydrochloric acid | HCl | Strong | Cleaning metals, bricks |
Phosphoric acid | H3PO4 | Moderate | Colas, rust removers |
Lactic acid | C3H6O3 | Weak | Yogurt, lotions |
Acetic acid | CH3COOH | Weak | Vinegar |
Boric acid | H3BO3 | Very weak | Eyewash, insecticide |
Hydrocyanic acid | HCN | Very weak | Plastics manufacture, toxic |
Name | Formula | Classification | Common Uses/Notes |
|---|---|---|---|
Sodium hydroxide | NaOH | Strong | Soap making, acid neutralization |
Potassium hydroxide | KOH | Strong | Liquid soap, biodiesel |
Lithium hydroxide | LiOH | Strong | Batteries |
Calcium hydroxide | Ca(OH)2 | Strong | Plaster, cement, soil neutralizer |
Magnesium hydroxide | Mg(OH)2 | Strong | Antacid, laxative |
Ammonia | NH3 | Weak | Cleaning, fertilizer |
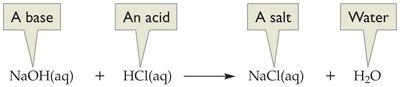
Neutralization
Neutralization is the reaction between an acid and a base, resulting in the formation of water and a salt. This process is fundamental in both laboratory and industrial chemistry.
General equation:
Limitations of the Arrhenius Theory
While useful, the Arrhenius theory has several limitations:
ions do not exist freely in water; they form hydronium ions ().
Does not explain the basicity of ammonia and similar compounds.
Applies only to aqueous solutions.
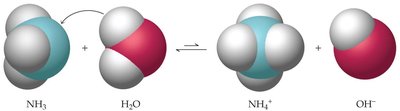
Brønsted-Lowry Theory
The Brønsted-Lowry theory expands the definition of acids and bases:
Acid: Proton donor.
Base: Proton acceptor.
Example reactions:

Salts
Salts are ionic compounds formed from cations other than hydrogen and anions other than hydroxide. They are often products of neutralization reactions and have various uses in medicine and industry.
Name | Formula | Uses |
|---|---|---|
Silver nitrate | AgNO3 | Germicide, antiseptic |
Stannous fluoride | SnF2 | Toothpaste additive |
Calcium sulfate | CaSO4 | Plaster casts |
Magnesium sulfate | MgSO4 | Laxative, foot baths |
Potassium permanganate | KMnO4 | Cauterizing agent, antiseptic |
Ferrous sulfate | FeSO4 | Treats anemia |
Zinc sulfate | ZnSO4 | Skin treatment |
Barium sulfate | BaSO4 | Contrast for X-rays |
Mercurous chloride | Hg2Cl2 | Laxative (no longer used) |
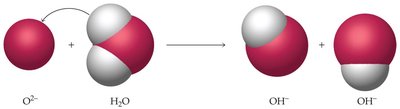
Anhydrides
Anhydrides are compounds formed by removing water from acids or bases. Nonmetal oxides are acidic anhydrides, while metal oxides are basic anhydrides.
Acidic anhydride example:
Basic anhydride example:
Strong and Weak Acids and Bases
Ionization in Water
Acids and bases are classified as strong or weak based on their degree of ionization in water.
Strong acids: Ionize completely in water. Example:
Weak acids: Ionize only partially. Example:
Strong bases: Dissociate completely. Example:
Weak bases: Ionize partially. Example:

Neutralization
Acid-Base Reactions
Neutralization reactions are essential for determining the amount of acid or base in a solution and for practical applications such as antacid use.
General equation:
Produces water and a salt.

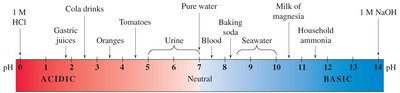
The pH Scale
Definition and Calculation
The pH scale quantifies the acidity or basicity of a solution, ranging from 0 (very acidic) to 14 (very basic).
pH formula:
pH 7 is neutral; values below 7 are acidic, above 7 are basic.
Relationship Between pH and Hydrogen Ion Concentration
pH | H+ Concentration (mol/L) |
|---|---|
0 | 1 × 100 |
1 | 1 × 10-1 |
2 | 1 × 10-2 |
... | ... |
14 | 1 × 10-14 |
Approximate pH Values of Common Solutions
Solution | pH |
|---|---|
Hydrochloric acid (4%) | 0 |
Gastric juice | 1.6–1.8 |
Soft drink | 2.0–4.0 |
Lemon juice | 2.1 |
Vinegar (4%) | 2.5 |
Urine | 5.5–7.0 |
Rainwater | 5.6 |
Saliva | 6.2–7.4 |
Milk | 6.3–6.6 |
Pure water | 7.0 |
Blood | 7.4 |
Fresh egg white | 7.6–8.0 |
Bile | 7.8–8.6 |
Milk of magnesia | 10.5 |
Washing soda | 12.0 |
Sodium hydroxide (4%) | 13.0 |
Buffers and Conjugate Acid–Base Pairs
Conjugate Acid–Base Pairs
Conjugate acid–base pairs differ by one proton. For example, chloride ion (Cl-) is the conjugate base of hydrochloric acid (HCl).
Buffer Solutions
Buffer solutions contain a weak acid and its conjugate base, allowing them to maintain a nearly constant pH when small amounts of acid or base are added.
Acid Rain
Formation and Effects
Acid rain is caused by nonmetal oxides in the air reacting with water to form acidic solutions. Rain with a pH less than 5.6 is considered acid rain. Major contributors include carbon dioxide (CO2), sulfur dioxide (SO2), nitrogen monoxide (NO), and nitrogen dioxide (NO2).
Antacids and Acid Reducers
Antacids: A Basic Remedy
Antacids are substances used to neutralize excess stomach acid and relieve symptoms of hyperacidity. Excessive use can lead to alkalosis, an increase in blood pH.
Substance | Formula | Product(s) |
|---|---|---|
Sodium bicarbonate | NaHCO3 | Baking soda |
Calcium carbonate | CaCO3 | Tums® |
Aluminum hydroxide | Al(OH)3 | Amphojel® |
Magnesium hydroxide | Mg(OH)2 | Milk of magnesia |

Acid Reducers
Acid reducers are medications that decrease acid production in the stomach, increasingly used instead of antacids.
Acids and Bases in Industry and at Home
Industrial and Household Uses
Acids and bases are widely used in industry and at home. Sulfuric acid is the most produced chemical in the U.S., used in fertilizers and batteries. Hydrochloric acid is used for cleaning and rust removal. Lime (CaO) is produced by heating limestone and is used in cement and soil treatment. Sodium hydroxide (lye) is used in soap making and cleaning. Ammonia is used in fertilizers and cleaning products.

Concentrated acids and bases are corrosive and hazardous to health, capable of denaturing proteins in living cells. The human body maintains proper pH in tissues, blood, and fluids through various mechanisms.
