 Back
BackLecture 18
Study Guide - Smart Notes

Acids and Bases: Theories and Environmental Examples
Arrhenius and Brønsted-Lowry Theories
Acids and bases are fundamental concepts in chemistry, with several theories describing their behavior. The Arrhenius theory defines acids as substances that produce H+ ions in water and bases as substances that produce OH- ions. The Brønsted-Lowry theory expands this definition, describing acids as proton (H+) donors and bases as proton acceptors.
Arrhenius Acid: Produces H+ in aqueous solution (e.g., HCl).
Arrhenius Base: Produces OH- in aqueous solution (e.g., NaOH).
Brønsted-Lowry Acid: Donates a proton (e.g., H2SO4).
Brønsted-Lowry Base: Accepts a proton (e.g., NH3).
Example: Ammonia (NH3) acts as a Brønsted-Lowry base by accepting a proton from water, forming NH4+ and OH-.
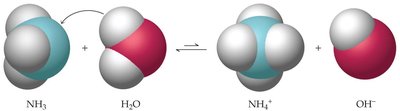
Environmental Example: Acid mine drainage, where sulfide minerals react to form sulfuric acid, is a real-world example of acid-base chemistry affecting ecosystems.

Neutralization Reactions
Acid-Base Neutralization
Neutralization reactions occur when an acid reacts with a base to produce water and a salt. These reactions are important in environmental and industrial processes.
General Equation:
Example: Sulfuric acid (H2SO4) reacts with sodium hydroxide (NaOH):
The pH Scale
Definition and Calculation
The pH scale is a logarithmic scale used to express the acidity or basicity of a solution. It is defined as the negative logarithm of the hydrogen ion concentration:
Formula:
Lower pH: More acidic (higher [H+])
Higher pH: More basic (lower [H+])
Each unit change: Represents a tenfold change in [H+]
Example: A solution with pH 4 has [H+] = M
Relationship Between pH and Hydrogen Ion Concentration
The concentration of hydrogen ions determines the pH of a solution. The following table shows the relationship:
pH | [H+] (mol/L) |
|---|---|
0 | 1 × 100 |
1 | 1 × 10-1 |
2 | 1 × 10-2 |
3 | 1 × 10-3 |
... | ... |
14 | 1 × 10-14 |
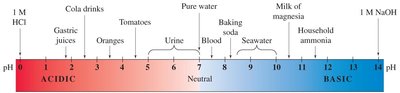
pH of Common Solutions
Different substances have characteristic pH values. The following table summarizes typical pH values:
Solution | pH |
|---|---|
Hydrochloric acid (4%) | 0 |
Gastric juice | 1.6–1.8 |
Soft drink | 2.0–4.0 |
Lemon juice | 2.1 |
Vinegar (4%) | 2.5 |
Urine | 5.5–7.0 |
Rainwater | 5.6 |
Saliva | 6.2–7.4 |
Milk | 6.3–6.6 |
Pure water | 7.0 |
Blood | 7.4 |
Fresh egg white | 7.6–8.0 |
Bile | 7.8–8.6 |
Milk of magnesia | 10.5 |
Washing soda | 12.0 |
Sodium hydroxide (4%) | 13.0 |
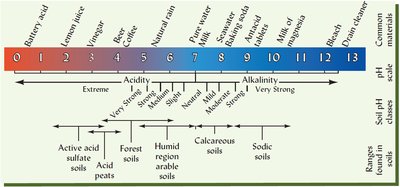
pH in Environmental Systems
pH as a Master Variable
pH controls many chemical and biological processes in soils, freshwater, and oceans. It affects nutrient availability, contaminant behavior, and biological activity.
Plant Growth: Most crops grow best at pH 6–7.5; low pH can cause toxicity (Al, Mn, H).
Microbial Activity: Fungi thrive at low pH; bacteria prefer neutral to high pH.
Contaminant Behavior: Metals are more soluble at low pH, affecting their bioavailability and toxicity.

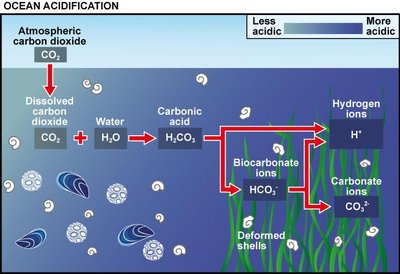
Carbonic Acid and Buffering Systems
Carbonic Acid Formation and Environmental Impact
Carbon dioxide dissolves in water to form carbonic acid, which plays a critical role in global and local environmental processes, including silicate weathering and soil pH regulation.
Formation:
Global Impact: Silicate rocks react with carbonic acid, consuming CO2 and affecting atmospheric levels.
Local Impact: Soil pH is lower near the surface due to biological activity and CO2 respiration.

Conjugate Acid–Base Pairs and Buffers
Conjugate acid–base pairs differ by one proton. Buffer solutions contain a weak acid and its conjugate base, allowing them to resist changes in pH when small amounts of acid or base are added.
Example: Bicarbonate (HCO3-) is the conjugate base of carbonic acid (H2CO3).
Buffer System: The carbonate buffer system is critical in oceans, freshwater, and high-pH soils.
Alkalinity and Environmental Chemistry
Definition and Importance
Alkalinity refers to the capacity of water to resist acidification, primarily due to carbonate and bicarbonate ions. It is related to, but not identical with, pH.
Higher pH: More basic, less acidic.
Alkalinity: Indicates buffering capacity against acidification.
Environmental Relevance: Oceans absorb about 30% of CO2 emissions, but their buffering capacity is not infinite.
Summary and Environmental Applications
Key Takeaways
Acid and base reactions regulate atmospheric CO2 levels, set conditions for agriculture, and allow oceans to serve as carbon sinks. pH is a master variable in environmental chemistry, affecting nutrient availability, contaminant behavior, and biological processes.
Acids & Bases: Fundamental to chemical reactions in nature.
pH Scale: Used to quantify acidity and basicity.
Buffers: Help maintain stable pH in environmental systems.
Alkalinity: Critical for resisting acidification in water bodies.
