 Back
BackLecture 17
Study Guide - Smart Notes

Acids & Bases
Acids and Bases in Earth Processes
Acid and base reactions are fundamental to many natural and human-driven processes. They regulate atmospheric CO2 levels, influence soil conditions for agriculture, and affect ocean chemistry.
Acidic soils are often indicated by red coloration and are common in rainforests, as seen in landslides in El Yunque National Forest, Puerto Rico.
Soil pH is a master variable for nutrient availability and agricultural productivity.
Acid-base reactions help oceans act as carbon sinks, but also contribute to risks like ocean acidification.

Acid and Base Theories
Two main theories describe acids and bases: Arrhenius and Brønsted-Lowry.
Arrhenius Theory:
Acid: A substance that ionizes in aqueous solution to produce hydrogen ions (H+).
Base: A substance that produces hydroxide ions (OH–) in aqueous solution.
Brønsted-Lowry Theory:
Acid: Proton donor.
Base: Proton acceptor.
Common Acids and Bases
Acids and bases are classified as strong or weak depending on their degree of ionization in water.
Name | Formula | Acid Strength | Common Uses/Notes |
|---|---|---|---|
Sulfuric acid | H2SO4 | Strong | Battery acid; ore processing; fertilizer manufacturing; oil refining |
Hydrochloric acid | HCl | Strong | Cleanin g metals and bricks; removing scale from boilers |
Phosphoric acid | H3PO4 | Moderate | Colas; rust removers |
Lactic acid | CH3CHOHCOOH | Weak | Yogurt; food additive; lotion additive |
Acetic acid | CH3COOH | Weak | Vinegar; food additive |
Boric acid | H3BO3 | Very weak | Antiseptic eyewash; roach poison |
Hydrocyanic acid | HCN | Very weak | Plastics manufacture; extremely toxic |
Name | Formula | Classification | Common Uses/Notes |
|---|---|---|---|
Sodium hydroxide | NaOH | Strong | Acid neutralization; soap making |
Potassium hydroxide | KOH | Strong | Liquid soap making; biodiesel fuels |
Lithium hydroxide | LiOH | Strong | Alkaline storage batteries |
Calcium hydroxide | Ca(OH)2 | Strong | Plaster; cement; soil neutralizer |
Magnesium hydroxide | Mg(OH)2 | Strong | Antacid; laxative |
Ammonia | NH3 | Weak | Household cleaner; fertilizer |
Acid-Base Reactions
Neutralization
When an acid reacts with a base, their properties are neutralized, producing water and a salt. This process is called neutralization.
General equation:
Example:
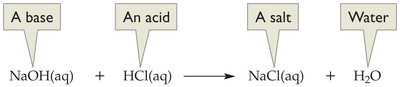
Neutralization reactions are used in titration to determine the concentration of acids or bases in solution.

Limitations of Arrhenius Theory
H+ ions do not exist freely in water; they form hydronium ions (H3O+).
Arrhenius theory does not explain the basicity of ammonia and similar compounds.
It applies only to aqueous solutions.
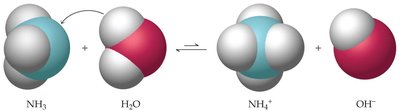
Brønsted-Lowry Theory
The Brønsted-Lowry theory expands the definition of acids and bases:
Acid: Proton donor
Base: Proton acceptor
Example: (when H2O is the base it recieves another proton)
Example: (when H2O is the acid, it forms OH)
Salts and Electrolytes
Formation and Properties of Salts
Salts are ionic compounds formed from the neutralization of acids and bases. They consist of cations other than hydrogen and anions other than hydroxide.
Examples: Epsom salts (MgSO4), NaCl, KCl, ZnSO4
Salts are concentrated in deserts by evaporation.


Electrolytes
Electrolytes are salts that conduct electricity when dissolved in water. Electrical conductivity measurements are used to determine salinity in water or soil.
High sodium soils may have elevated salts but are not always saline.
Salt marsh soils can have very high salinity.

Strong and Weak Acids and Bases
Ionization in Water
Acids and bases are classified as strong or weak based on their ionization in water.
Strong acids: Ionize completely in water (e.g., HCl).
Weak acids: Ionize only partially (e.g., HCN).
Strong bases: Dissociate completely (e.g., NaOH).
Weak bases: Ionize only partially (e.g., NH3).
The pH Scale
Definition and Calculation
The pH scale expresses the acidity or basicity of a solution. It is defined as:
pH:
pH ranges from 0 (very acidic) to 14 (very basic).
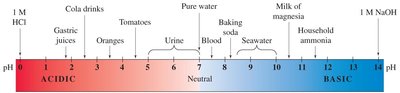
Solution | pH |
|---|---|
Hydrochloric acid (4%) | 0 |
Gastric juice | 1.6–1.8 |
Soft drink | 2.0–4.0 |
Lemon juice | 2.1 |
Vinegar (4%) | 2.5 |
Urine | 5.5–7.0 |
Rainwater | 5.6 |
Saliva | 6.2–7.4 |
Milk | 6.3–6.6 |
Pure water | 7.0 |
Blood | 7.4 |
Fresh egg white | 7.6–8.0 |
Bile | 7.8–8.6 |
Milk of magnesia | 10.5 |
Washing soda | 12.0 |
Sodium hydroxide (4%) | 13.0 |
Carbonic Acid and Global Processes
Carbonation Reactions
Carbon dioxide dissolves in water to form carbonic acid, which plays a key role in global carbon cycling and soil pH regulation.
CO2 (g) → CO2 (aq)
CO2 (aq) + H2O → H2CO3 (aq)
Silicate rocks react with carbonic acid to form carbonate/bicarbonate and cations, contributing to global CO2 consumption.
Soil pH is lower near the surface due to biological activity and CO2 respiration from roots and microbes.
Summary
Acids and bases are defined by two main theories: Arrhenius and Brønsted-Lowry.
Neutralization reactions produce water and salts.
The pH scale quantifies acidity and basicity.
Acid-base chemistry is central to environmental and biological processes.
