 Back
BackAir Chemistry: Structure, Composition, Pollution, and Environmental Impact
Study Guide - Smart Notes

Air Chemistry and Earth's Atmosphere
Atmospheric Structure and Composition
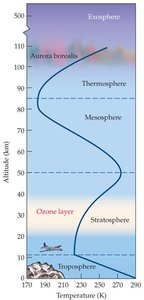
The Earth's atmosphere is divided into several distinct layers, each with unique properties and functions. Understanding these layers is essential for grasping the chemistry of air and its environmental implications.
Troposphere: The lowest layer, containing most living organisms. Temperature decreases with altitude.
Stratosphere: Above the troposphere, contains the ozone layer. Temperature increases with altitude.
Mesosphere: Above the stratosphere, where temperatures decrease again.
Thermosphere: The uppermost layer, with increasing temperatures due to solar radiation.
The composition of clean, dry air near sea level is primarily nitrogen and oxygen, with trace amounts of other gases.
Component | Percent by Volume |
|---|---|
Nitrogen (N2) | 78.08 |
Oxygen (O2) | 20.94 |
Argon (Ar) | 0.93 |
Carbon dioxide (CO2) | 0.04 |
Trace gases | <0.01 |
Nitrogen Cycle and Atmospheric Chemistry
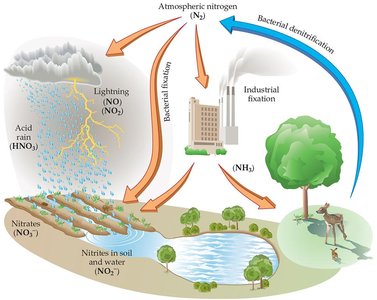
Nitrogen is the most abundant atmospheric gas, but most organisms cannot use it directly. It must be "fixed"—combined with other elements—before it becomes biologically available.
Natural fixation: Lightning converts N2 and O2 into nitrogen oxides, which form nitric acid in rain.
Industrial fixation: The Haber–Bosch process produces ammonia (NH3) from nitrogen and hydrogen.
Bacterial fixation: Certain bacteria in plant roots convert atmospheric nitrogen into usable forms.
Example: Lightning fixes nitrogen as follows:
Air Pollution and Smog
Pollution Goes Global
A pollutant is any substance found in the wrong place at the wrong time. Urbanization and globalization have made air pollution a worldwide concern.
Industrial Smog
Industrial smog results from the burning of coal and other industrial activities, producing a mixture of smoke and fog.

Oxides of Carbon: Burning coal produces carbon dioxide, carbon monoxide, and soot.
Oxides of Sulfur: Sulfur in coal forms sulfur dioxide and sulfur trioxide, which react with water to produce sulfuric acid.
Particulate Matter (PM): Solid and liquid particles, including unburned minerals, contribute to air pollution.
Types of Smog
Smog can be classified into two main types:
Industrial Smog | Photochemical Smog | |
|---|---|---|
Alternative names | Winter smog; sulfurous smog; "London smog" | Summer smog; "Los Angeles smog" |
Main initial components | SO2; particulates | NOx; hydrocarbons |
Secondary components | SO3; H2SO4 | O3; aldehydes; PAN |
Main sources | Power plants; factories | Automobiles |
Typical weather | Cold; damp; foggy | Warm; dry; sunny |
Health and Environmental Effects of Industrial Smog
Health: Sulfuric acid and particulates damage lung tissue, leading to pulmonary emphysema.
Environmental: Acidic precipitation and particulates harm plants and crops.
Controlling Industrial Smog
Several technologies are used to reduce industrial smog:
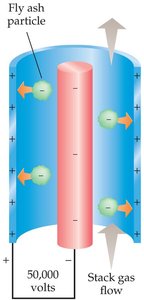
Electrostatic precipitators: Induce charges on particulates, attracting them to plates for removal.
Bag filtration: Removes particulates using filters.
Cyclone separators: Use spiral motion to deposit heavier particulates.
Wet scrubbers: Pass gases through water to remove particulates.
Sulfur removal: Sulfur can be removed from coal or neutralized with limestone.
Automobile Emissions and Air Chemistry
Combustion of Gasoline
Gasoline is a mixture of hydrocarbons, with octane (C8H18) as a representative compound. Complete combustion produces carbon dioxide and water:
Carbon Monoxide
Incomplete combustion produces carbon monoxide (CO), a toxic, colorless, odorless gas. CO binds to hemoglobin, preventing oxygen transport in the blood.
Nitrogen Oxides
High temperatures in engines cause nitrogen and oxygen to react, forming nitrogen oxides (NOx):
NOx reacts with water to form acids, contributing to smog and acid rain. High concentrations can cause lung damage.
Volatile Organic Compounds (VOCs)
VOCs are hydrocarbons released from gasoline, exhaust, paints, and consumer products. They react with oxygen or ozone to form aldehydes and contribute to smog formation.
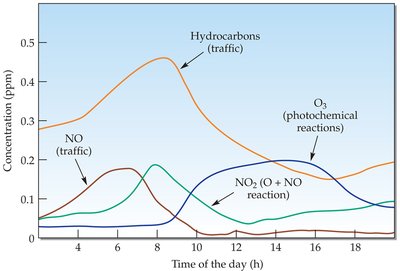
Photochemical Smog
Photochemical smog forms when pollutants react with sunlight, producing an amber haze. Pollutant concentrations vary throughout the day.
Solutions to Photochemical Smog
Reduce pollutant emissions through improved gasoline storage and catalytic converters.
Acid Rain
Formation and Effects
Acid rain is precipitation with a pH less than 5.6, formed when sulfur and nitrogen oxides react with water to produce acids.
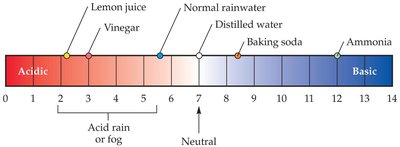
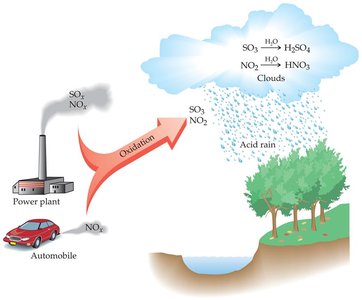
Acid rain damages natural and manufactured environments, harming plants, aquatic life, and infrastructure.
Indoor Air Pollution
Sources and Health Effects
Indoor air pollution can be much higher than outdoor levels. Common sources include gas stoves, cigarette smoke, kerosene heaters, mold, and ozone from electronic devices.
Cigarette smoke: Contains carcinogens and increases risk of heart disease, lung cancer, miscarriage, and SIDS.
Radon: A radioactive noble gas released from rocks and soil, decays by alpha emission, and poses health risks.
Ozone: The Double-Edged Sword
Ozone in the Atmosphere
Ozone (O3) is an allotrope of oxygen. In the troposphere, it is a pollutant; in the stratosphere, it protects life by absorbing ultraviolet radiation.
UV radiation splits O2 molecules into oxygen atoms:

Oxygen atoms react with O2 to form ozone:
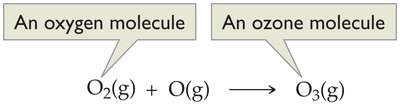
Ozone absorbs UV radiation, protecting living organisms.
Ozone Depletion
Chlorofluorocarbons (CFCs) destroy ozone in the stratosphere, leading to the ozone hole, especially over Antarctica.
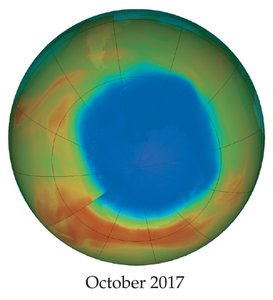
Many countries have banned CFCs, and substitutes have been developed.
Carbon Dioxide and Climate Change
Greenhouse Effect
Carbon dioxide (CO2) is a greenhouse gas that traps heat in the atmosphere, contributing to global warming.
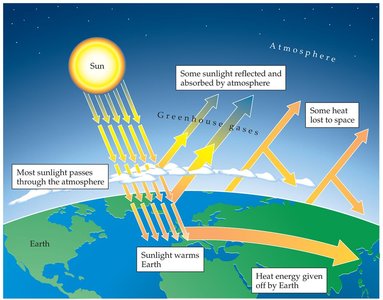
Human activities have increased atmospheric CO2, leading to rising global temperatures.
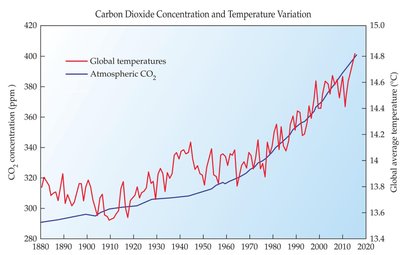
Climate Change and Mitigation
Climate change impacts food production, flooding, and disease. Mitigation requires reducing greenhouse gas emissions through technologies like solar, nuclear, wind, and carbon sequestration.
