 Back
BackCH100 Exam 2 Study Guide: Molecules, Compounds, Chemical Composition, Reactions, and Quantities
Study Guide - Smart Notes

Chapter 5: Molecules and Compounds
Formation and Properties of Compounds
When elements combine to form compounds, their properties change completely. The Law of Constant Composition states that all samples of a given compound have the same proportions of their constituent elements. For example, water is always H2O; changing the number of hydrogen or oxygen atoms would result in a different compound. The mass ratio of oxygen to hydrogen in water is always 8:1, calculated from their atomic masses (O = 16, H = 1).
Polyatomic Ions and Chemical Formulas
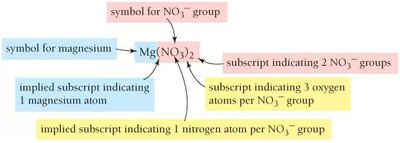
Polyatomic ions are groups of atoms that act as a single ion in chemical formulas. Parentheses are used in formulas when there are multiples of a polyatomic ion, such as in Mg(NO3)2. It is important to determine the number of each atom in a formula and understand the use of parentheses.
Example: In Mg(NO3)2:
Mg: 1 atom
NO3: 2 groups
N: 2 atoms (1 per group × 2 groups)
O: 6 atoms (3 per group × 2 groups)
Molecular vs. Empirical Formulas
The molecular formula shows the exact number of atoms of each element in a molecule, while the empirical formula shows the simplest whole-number ratio of the elements.
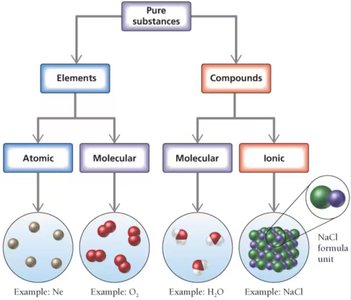
Classification of Elements and Compounds
Elements and compounds can be classified as atomic, molecular, or ionic. Atomic elements exist as single atoms (e.g., Ne), molecular elements exist as molecules (e.g., O2), molecular compounds are composed of molecules (e.g., H2O), and ionic compounds consist of ions (e.g., NaCl).
Order of Elements in Chemical Formulas
Generally, the less electronegative element is written first in a chemical formula. In organic compounds, carbon is written first, followed by hydrogen, then other elements.
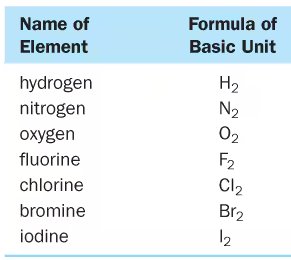
Diatomic Molecules
Diatomic molecules are molecules composed of two atoms of the same element. Common diatomic elements in nature include hydrogen, nitrogen, oxygen, fluorine, chlorine, bromine, and iodine.
Name of Element | Formula of Basic Unit |
|---|---|
hydrogen | H2 |
nitrogen | N2 |
oxygen | O2 |
fluorine | F2 |
chlorine | Cl2 |
bromine | Br2 |
iodine | I2 |
Chapter 6: Chemical Composition
Avogadro’s Number and the Mole
Avogadro’s number (6.022 × 1023) is the number of atoms in one mole of any substance. The mole is a fundamental unit for counting atoms, molecules, or ions in chemistry.
Molar Mass
The molar mass is the mass of one mole of atoms of an element, numerically equal to the atomic mass but expressed in grams. For compounds, the molar mass is the sum of the molar masses of the constituent elements.
Example: For CO2:
C: 12.01 g/mol
O: 16.00 g/mol × 2 = 32.00 g/mol
Total: 44.01 g/mol
Chapter 7: Chemical Reactions
Types of Chemical Reactions
Chemical reactions can be classified into several types:
Oxidation-Reduction: Involves transfer of electrons.
Combustion: Rapid reaction with oxygen producing heat and light.
Precipitation: Formation of a solid from solution.
Gas Evolution: Production of a gas during the reaction.
Evidence of Chemical Reactions
Evidence that a chemical reaction has occurred includes color change, formation of a precipitate, gas production, and energy change (heat/light).
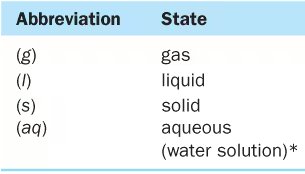
States of Reactants and Products
Abbreviations are used to indicate the physical state of substances in chemical equations:
Abbreviation | State |
|---|---|
(g) | gas |
(l) | liquid |
(s) | solid |
(aq) | aqueous (water solution) |
Balancing Chemical Equations
Balancing chemical equations ensures the number of each type of atom is equal on both sides. This is achieved by adjusting coefficients, not subscripts. The process changes the number of molecules but not their identity.
Aqueous Solutions
When compounds dissolve in water, they form aqueous solutions where ions are separated and move independently.
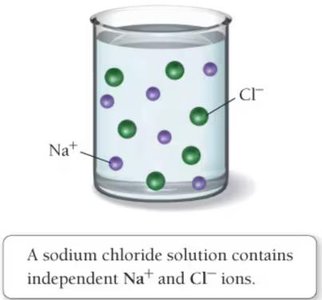
Molecular, Complete Ionic, and Net Ionic Equations
Molecular equation: Shows all reactants and products as compounds.
Complete ionic equation: Shows all ions present in solution.
Net ionic equation: Shows only the ions and molecules directly involved in the reaction.
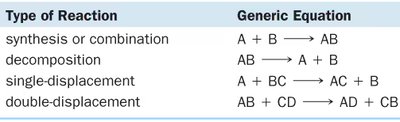
Classification of Reactions by Atom Movement
Chemical reactions can be classified based on how atoms are rearranged:
Type of Reaction | Generic Equation |
|---|---|
synthesis or combination | A + B → AB |
decomposition | AB → A + B |
single-displacement | A + BC → AC + B |
double-displacement | AB + CD → AD + CB |
Chapter 8: Quantities in Chemical Reactions
Stoichiometry
Stoichiometry is the numerical relationship between chemical quantities in a balanced equation. It allows prediction of product amounts from reactants and determination of required reactant quantities.
Limiting Reactant and Theoretical Yield
The limiting reactant is the reactant that is completely consumed first, limiting the amount of product formed. The theoretical yield is the maximum amount of product that can be formed from the given reactants.
Analogy: Making Pancakes
Stoichiometry can be understood using a recipe analogy. For example, if 1 cup flour + 2 eggs + ½ tsp baking powder makes 5 pancakes, the ingredient that runs out first limits the number of pancakes (limiting reactant), and the maximum possible pancakes is the theoretical yield.

Enthalpy and Reaction Types
Enthalpy is the heat content of a system. Endothermic reactions absorb heat, while exothermic reactions release heat.
Endothermic: Energy is absorbed; surroundings feel cooler.
Exothermic: Energy is released; surroundings feel warmer.
Additional info: The notes do not cover calculation of formula mass, naming compounds, mass-to-mass conversions, or percent yield for this exam.
