 Back
BackChapter 11: Gases – Properties, Laws, and Applications
Study Guide - Smart Notes

Gases: Properties and Behavior
Introduction to Gases
Gases are one of the fundamental states of matter, characterized by their ability to expand and fill any container. Understanding gases is essential for explaining phenomena such as breathing, weather, and chemical reactions involving gases.
How Straws Work: Atmospheric Pressure and Gas Behavior
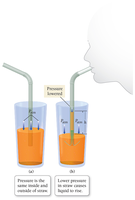
Pressure Differences and Drinking with Straws
When drinking from a straw, removing air from inside the straw creates a pressure difference between the inside and outside. Atmospheric pressure pushes the liquid up the straw. The maximum height a liquid can be pushed by atmospheric pressure is about 10.3 meters for water, due to the balance between atmospheric pressure and the weight of the liquid column.


Kinetic Molecular Theory: A Model for Gases
Postulates of the Kinetic Molecular Theory
The kinetic molecular theory explains the behavior of gases by modeling them as a collection of particles in constant, straight-line motion. The main assumptions are:
Gas particles are in constant, random motion.
There are no attractive or repulsive forces between particles.
The volume of the particles is negligible compared to the space between them.
The average kinetic energy of the particles is proportional to the temperature in kelvins.

Properties of Gases Explained by Kinetic Molecular Theory
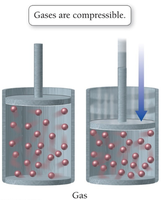
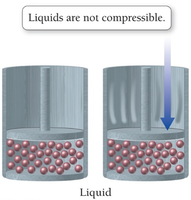
Compressibility: Gases are compressible because of the large amount of empty space between particles.
Shape and Volume: Gases assume the shape and volume of their container due to negligible intermolecular attractions.
Low Density: Gases have much lower densities than liquids and solids because of the large distances between particles.

Pressure: The Result of Molecular Collisions
Definition and Origin of Pressure
Pressure is defined as the force exerted per unit area by gas molecules as they collide with surfaces. The SI unit of pressure is the pascal (Pa), but atmospheres (atm) and millimeters of mercury (mm Hg) are also commonly used.

Pressure and Altitude
As altitude increases, atmospheric pressure decreases due to fewer air molecules per unit volume. This can cause discomfort, such as ear pain, due to pressure imbalances.
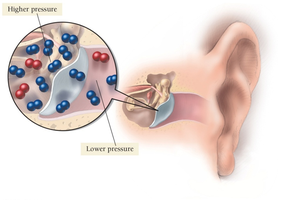
Factors Affecting Pressure
Increasing the number of gas particles in a fixed volume increases pressure.
Decreasing the number of particles decreases pressure.

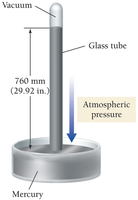
Units of Pressure
Common Units and Conversions
Atmosphere (atm): Average pressure at sea level.
Pascals (Pa): SI unit; 1 atm = 101,325 Pa.
Millimeters of mercury (mm Hg): Based on the height of a mercury column; 1 atm = 760 mm Hg.
Torr: 1 torr = 1 mm Hg.

Gas Laws
Boyle’s Law: Pressure and Volume
Boyle’s law states that the volume of a gas is inversely proportional to its pressure at constant temperature and amount of gas.
Equation:
As volume decreases, pressure increases, and vice versa.
Charles’s Law: Volume and Temperature
Charles’s law states that the volume of a gas is directly proportional to its temperature (in kelvins) at constant pressure and amount of gas.
Equation:
As temperature increases, volume increases.
Avogadro’s Law: Volume and Moles
Avogadro’s law states that the volume of a gas is directly proportional to the number of moles at constant temperature and pressure.
Equation:
The Combined Gas Law
The combined gas law relates pressure, volume, and temperature for a fixed amount of gas:
The Ideal Gas Law
The ideal gas law combines all the simple gas laws into one equation:
P = pressure (atm)
V = volume (L)
n = moles of gas
R = ideal gas constant (0.0821 L·atm/mol·K)
T = temperature (K)
Partial Pressures and Gas Mixtures
Dalton’s Law of Partial Pressures
In a mixture of gases, each gas exerts its own pressure, called partial pressure. The total pressure is the sum of the partial pressures of all components.
Equation:
The partial pressure of a component is its fractional composition times the total pressure.
Applications and Environmental Chemistry
Air Pollution
Major gaseous air pollutants include sulfur dioxide, carbon monoxide, ozone, and nitrogen dioxide. These pollutants can cause respiratory problems and environmental damage. Legislation such as the Clean Air Act has reduced pollutant levels in many cities.
Summary Table: Common Units of Pressure
Unit | Symbol | Equivalent to 1 atm |
|---|---|---|
Atmosphere | atm | 1 atm |
Millimeter of mercury | mm Hg | 760 mm Hg |
Torr | torr | 760 torr |
Pounds per square inch | psi | 14.7 psi |
Pascals | Pa | 101,325 Pa |
Key Equations
Boyle’s Law:
Charles’s Law:
Avogadro’s Law:
Combined Gas Law:
Ideal Gas Law:
Dalton’s Law:
Learning Objectives
Convert between different units of pressure.
Apply simple gas laws to solve problems.
Use the combined and ideal gas laws for calculations involving gases.
Relate total and partial pressures in mixtures of gases.
Calculate stoichiometry for gases in chemical reactions.
