 Back
BackChapter 13: Solutions – Structure, Properties, and Applications
Study Guide - Smart Notes

Solutions: Homogeneous Mixtures
Definition and Examples
Solutions are homogeneous mixtures composed of two or more substances. The component present in the largest amount is called the solvent, while the other components are called solutes. Solutions can be found in all states of matter and are common in everyday life, such as seawater, blood plasma, and carbonated beverages.
Solvent: The substance present in the greatest amount; in aqueous solutions, water is the solvent.
Solute: The substance(s) present in lesser amounts and dissolved by the solvent.
Solutions can be solid/liquid, gas/liquid, liquid/liquid, or other combinations.

How Substances Dissolve
Solubility and Intermolecular Forces
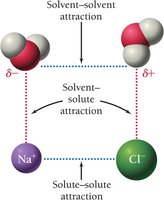
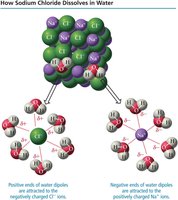
The process of dissolving involves competition between solute-solute, solvent-solvent, and solvent-solute attractions. A solute will dissolve if the solvent-solute attractions are strong enough to overcome the other forces. Water, being a polar molecule, is especially effective at dissolving ionic and other polar substances.
"Like dissolves like": Polar solvents dissolve polar solutes; nonpolar solvents dissolve nonpolar solutes.
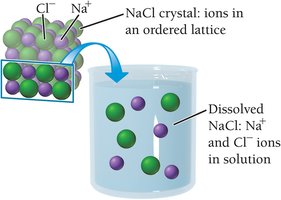
When ionic compounds like NaCl dissolve in water, the positive and negative ions are surrounded by water molecules and dispersed throughout the solution.
Solubility and Saturation
Types of Solutions
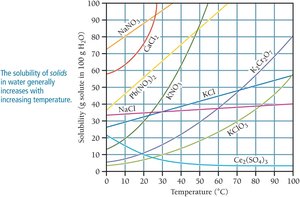
Solubility is the maximum amount of solute that can dissolve in a given amount of solvent at a specific temperature. Solutions can be classified based on their solute content relative to solubility:
Saturated solution: Contains the maximum amount of solute that can dissolve under the given conditions. Additional solute will not dissolve.
Unsaturated solution: Contains less than the maximum amount of solute; more solute can dissolve.
Supersaturated solution: Contains more solute than is normally possible at that temperature; excess solute will precipitate out.
Solubility of solids generally increases with temperature, while the solubility of gases decreases with increasing temperature but increases with pressure.
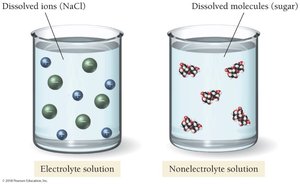
Electrolyte and Nonelectrolyte Solutions
Electrical Conductivity
Solutions can be classified based on their ability to conduct electricity:
Electrolyte solutions: Contain dissolved ions and conduct electricity (e.g., NaCl in water).
Nonelectrolyte solutions: Contain dissolved molecules and do not conduct electricity (e.g., sugar in water).
Solution Concentration
Mass Percent, Molarity, and Molality
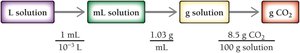
Concentration quantifies the amount of solute in a given amount of solution. Common units include:
Mass percent (%):
Molarity (M):
Molality (m):


Solution Preparation and Dilution
Making Solutions of Desired Concentration
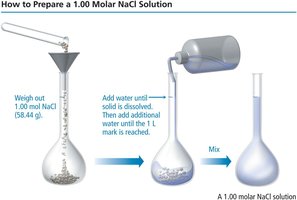
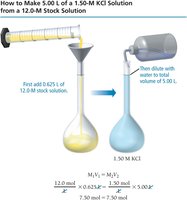
To prepare a solution of specific molarity, dissolve the required amount of solute in a volumetric flask and add solvent up to the desired volume. To dilute a stock solution, use the dilution equation:
Where and are the molarity and volume of the concentrated solution, and and are those of the diluted solution.
Solution Stoichiometry
Using Concentration in Chemical Reactions
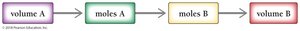
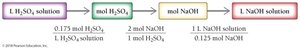
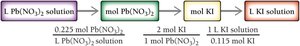
In reactions involving solutions, the volume and concentration can be used to determine the amount of reactants or products. The general approach is:
Convert volume to moles using molarity.
Use stoichiometric coefficients from the balanced equation to relate moles of different substances.
Convert moles back to volume or mass as needed.
Colligative Properties
Freezing Point Depression and Boiling Point Elevation
Colligative properties depend only on the number of solute particles in solution, not their identity. Adding a nonvolatile solute to a solvent lowers the freezing point and raises the boiling point of the solution.
Freezing point depression:
Boiling point elevation:
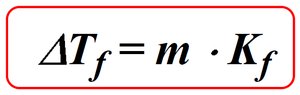
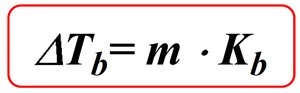
Osmosis and Osmotic Pressure
Movement of Solvent Across Membranes
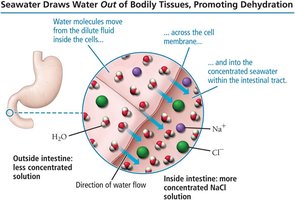
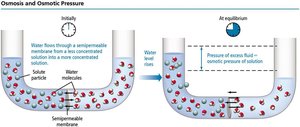
Osmosis is the flow of solvent from a less concentrated solution to a more concentrated one through a semipermeable membrane. The pressure required to stop this flow is called osmotic pressure. Osmosis is crucial in biological systems and medical applications.
Hyperosmotic solution: Higher osmotic pressure than bodily fluids; draws water out of cells.
Hypoosmotic solution: Lower osmotic pressure than bodily fluids; causes water to enter cells.
Isoosmotic solution: Same osmotic pressure as bodily fluids; no net movement of water.
Summary Table: Common Types of Solutions
Solute State | Solvent State | Example |
|---|---|---|
Gas | Liquid | Carbonated water (CO2 in water) |
Liquid | Liquid | Alcohol in water |
Solid | Liquid | Salt in water |
Gas | Solid | Hydrogen in palladium |
Solid | Solid | Alloys (e.g., brass) |
Key Equations
Mass percent:
Molarity:
Molality:
Freezing point depression:
Boiling point elevation:
Dilution:
Learning Objectives
Define solution, solute, and solvent.
Relate solubility of solids and gases to temperature and pressure.
Calculate mass percent, molarity, and molality.
Use concentration units in calculations and stoichiometry.
Explain and calculate colligative properties and osmosis.
