 Back
BackChemical Composition and Quantitative Relationships in Chemistry
Study Guide - Smart Notes

Chemical Composition
Introduction to Chemical Composition
Chemical composition refers to the types and amounts of elements that make up a substance. Understanding chemical composition is essential for quantifying substances in chemical reactions and for converting between mass, moles, and number of particles.
Converting Between Grams and Moles of an Element
Understanding the Mole Concept
Mole (mol): The SI unit for amount of substance, defined as containing exactly entities (Avogadro's number).
Molar Mass: The mass of one mole of a substance, typically expressed in grams per mole (g/mol).
The lighter the atom, the less mass in 1 mol of atoms; the lighter the atom, the more atoms there are in 1 g.
Example: Calculate the moles of carbon in 0.0265 g of pencil lead (pure carbon).
Given:
Calculation:
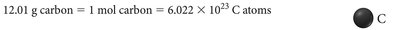
Converting Between Grams and Number of Atoms
Multi-Step Conversions
To convert grams of an element to number of atoms, use the following steps:
Grams of element → Moles of element → Number of atoms
Example: How many copper atoms are in a penny weighing 3.10 g (pure Cu)?
Given: , atoms
Calculation: atoms Cu

Counting Molecules by the Gram
Molar Mass of Compounds
Molar Mass (of a compound): The mass of one mole of molecules or formula units of a compound.
For compounds, add the molar masses of all atoms in the formula.
Example: Molar mass of :
2 mol H:
1 mol O:
Total:
Converting Between Grams and Moles of a Compound
Application to Compounds
To find moles from grams:
To find grams from moles:
Example: Find the number of moles in a 22.5 g sample of dry ice ():
Molar mass
Converting Between Grams of a Compound and Number of Molecules
Multi-Step Conversion
Grams of compound → Moles of compound → Number of molecules
Example: Find the number of molecules in 22.5 g of dry ice:
molecules

Converting Between Number of Molecules and Mass of a Compound
Reverse Multi-Step Conversion
Number of molecules → Moles of compound → Grams of compound
Example: What is the mass of molecules?
Molar mass

Chemical Formulas as Conversion Factors
Understanding Relationships in Chemical Formulas
Chemical formulas indicate the ratio of elements in a compound.
These ratios can be used as conversion factors between moles of compound and moles of constituent elements.
Example: In , 1 mol $H_2O$ contains 2 mol H and 1 mol O.
To find moles of H in 50 mol :
Converting Between Moles of a Compound and Moles of a Constituent Element
Application to Compounds
Use the subscripts in the chemical formula as conversion factors.
Example: Determine the number of moles of O in 3.7 mol of :
contains 3 O atoms per formula unit.

Converting Between Grams of a Compound and Grams of a Constituent Element
Multi-Step Conversion
Grams of compound → Moles of compound → Moles of element → Grams of element
Example: Determine the mass of carbon in 55.4 g of :
Molar mass
10 mol C per 1 mol

Mass Percent Composition of Compounds
Definition and Calculation
Mass percent composition is the percentage by mass of each element in a compound.
Formula:
Example: A 0.358 g sample of chromium reacts with oxygen to form 0.523 g of metal oxide. What is the mass percent of chromium?

Mass Percent Composition as a Conversion Factor
Using Mass Percent in Calculations
Mass percent can be used as a conversion factor between the mass of an element and the mass of the compound.
For example, if a compound is 39% Na by mass, then 100 g of the compound contains 39 g Na.
Example: Find the mass of NaCl that contains 2.4 g of Na (39% Na):
Mass Percent Composition from a Chemical Formula
Calculation from Formula
Mass percent can be determined from the chemical formula using molar masses.
Formula:
Example: Find the mass percent of Cl in Freon-112 ():
Molar mass Cl = 35.45 g/mol, 4 Cl atoms
Molar mass
Conversion Factors from Chemical Formulas
Using Chemical Formulas for Quantitative Relationships
Chemical formulas provide relationships between numbers of atoms, moles, and masses.
These relationships are essential for determining the amounts of constituent atoms or molecules in a given sample.
Practice and Application
Worked Examples
How many moles of are in 4.49 g?
How many moles of P?
How many moles of O?
Summary Table: Key Conversion Factors
Quantity | Conversion Factor | Example |
|---|---|---|
Grams → Moles | ||
Moles → Grams | ||
Moles → Molecules | ||
Molecules → Moles | ||
Mass % → Mass |
