 Back
BackChemical Composition: Formula Mass, the Mole, and Solution Concentrations
Study Guide - Smart Notes

Formula Mass and the Mole Concept
Definition of Formula Mass
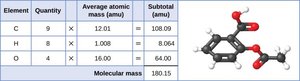
The formula mass of a substance is the sum of the average atomic masses of all the atoms in the chemical formula. For covalent substances, this is also called the molecular mass. For ionic compounds, the term "formula mass" is preferred, as these substances do not exist as discrete molecules.
Covalent substances exist as discrete molecules; their formula mass is the sum of the atomic masses of all atoms in the molecule.
Ionic compounds are composed of cations and anions in fixed ratios; their formula mass is the sum of the atomic masses of the ions in the empirical formula.
The unit for formula mass is atomic mass units (amu).
Example: The molecular mass of aspirin (C9H8O4) is calculated as follows:
The Mole and Avogadro’s Number
The mole is a counting unit in chemistry, analogous to terms like dozen or pair. One mole contains exactly 6.022 × 1023 entities (Avogadro’s number, NA), which may be atoms, molecules, or ions.
1 mole of any substance contains 6.022 × 1023 particles.
The molar mass (g/mol) of a substance is numerically equal to its formula mass in amu, but expressed in grams per mole.
Example: 1 mole of carbon-12 atoms has a mass of 12 g.
Relating Mass, Moles, and Number of Particles
Conversions between mass, moles, and number of particles are fundamental in chemistry. The following relationships are used:
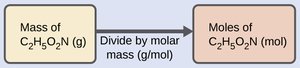
To convert mass to moles: divide by molar mass.
To convert moles to mass: multiply by molar mass.
To convert moles to number of particles: multiply by Avogadro’s number.
To convert number of particles to moles: divide by Avogadro’s number.
Example: Calculating moles of potassium from mass.

Example: Calculating mass of argon from moles.

Example: Calculating number of copper atoms from mass.

Example: Calculating moles of a compound from mass (e.g., C2H5O2N).
Example: Calculating mass of vitamin C from moles.

Example: Calculating number of saccharin molecules from mass.

Determining Empirical and Molecular Formulas
Percent Composition
Percent composition is the percentage by mass of each element in a compound. It can be determined experimentally or calculated from the chemical formula.
Percent composition = (mass of element in 1 mol of compound / molar mass of compound) × 100%
Example: For NH3, calculate the percent composition of N and H using their atomic masses and the formula mass of NH3.
Empirical Formula Determination
The empirical formula represents the simplest whole-number ratio of elements in a compound. It can be determined from the masses or percent composition of the elements.
Convert masses (or percentages) of elements to moles using molar masses.
Divide each mole value by the smallest number of moles calculated.
If necessary, multiply all ratios by an integer to obtain whole numbers.
Example: A compound contains 1.71 g C and 0.287 g H. Convert to moles, find the ratio, and write the empirical formula.
Molecular Formula Determination
The molecular formula gives the actual number of atoms of each element in a molecule. It is a whole-number multiple of the empirical formula.
n = (molar mass of compound) / (empirical formula mass)
Molecular formula = (empirical formula) × n
Example: If empirical formula is CH2O (mass = 30 amu) and molecular mass is 180 amu, n = 180/30 = 6, so molecular formula is C6H12O6.
Molarity and Solution Concentrations
Definition of Molarity
Molarity (M) is the number of moles of solute per liter of solution. It is a common unit for expressing solution concentration.
Example: A 355 mL soft drink contains 0.133 mol sucrose. Molarity = 0.133 mol / 0.355 L = 0.375 M.
Dilution of Solutions
Dilution is the process of decreasing the concentration of a solution by adding more solvent. The amount of solute remains constant before and after dilution.
The dilution equation:
Where M1 and V1 are the initial molarity and volume, and M2 and V2 are the final molarity and volume.
Example: If 0.850 L of 5.00 M Cu(NO3)2 is diluted to 1.80 L, the new molarity is .
Other Units for Solution Concentrations
Mass Percentage
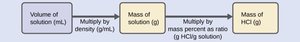
Mass percentage expresses the mass of a component as a percentage of the total solution mass.
Mass % = (mass of component / mass of solution) × 100%
Example: Calculating mass percent of HCl in a solution.
Volume Percentage and Mass-Volume Percentage
Volume percentage is used when both solute and solvent are liquids. Mass-volume percentage is common in medical and biological contexts, such as saline solutions.
Mass-volume % = (mass of solute in g / volume of solution in mL) × 100%
Example: Physiological saline is 0.9% (m/v), meaning 0.9 g NaCl per 100 mL solution.
Parts per Million (ppm) and Parts per Billion (ppb)
Very low concentrations are often expressed as ppm or ppb, especially in environmental and analytical chemistry.
ppm = (mass of solute / mass of solution) × 106
ppb = (mass of solute / mass of solution) × 109
