 Back
BackChemical Quantities in Reactions: A Study Guide
Study Guide - Smart Notes

Chapter 9: Chemical Quantities in Reactions
9.1 Conservation of Mass
The Law of Conservation of Mass states that in an ordinary chemical reaction, matter cannot be created or destroyed. This means the total mass of the reactants equals the total mass of the products in a chemical reaction.
Definition: The mass of the products is always equal to the mass of the reactants.
Application: This law is fundamental for balancing chemical equations and for quantitative chemical calculations.
Example: The reaction of silver (Ag) with sulfur (S) to form silver sulfide (Ag2S) demonstrates that the combined mass of Ag and S before the reaction equals the mass of Ag2S after the reaction.

Table 9.1 summarizes the information available from a balanced equation, including the number of atoms, moles, and mass relationships.

Sample Problem: The combustion of methane (CH4) with oxygen produces carbon dioxide and water. The total mass of reactants equals the total mass of products, confirming the law of conservation of mass.

9.2 Mole Relationships in Chemical Equations
Chemical equations provide the mole ratios of reactants and products, which are essential for quantitative chemical calculations. A mole–mole factor is a ratio derived from the coefficients of a balanced equation, relating the moles of any two substances.
Definition: A mole–mole factor is a conversion factor between the moles of two substances in a balanced chemical equation.
Example: In the reaction 4Fe(s) + 3O2(g) → 2Fe2O3(s), the mole–mole factor between O2 and Fe2O3 is 3:2.
Application: Used to calculate the amount of one substance that reacts with or is produced from a given amount of another substance.


Sample Calculation: To find how many moles of Fe2O3 can form from 6.0 moles of O2:
Equation: 4Fe(s) + 3O2(g) → 2Fe2O3(s)
Relationship: 3 mol O2 = 2 mol Fe2O3
Calculation:
9.3 Mass Calculations for Chemical Reactions
To determine the mass of a product or reactant in a chemical reaction, use the following steps:
Convert grams of known substance to moles (using molar mass).
Use the mole–mole factor from the balanced equation to convert moles of known to moles of unknown.
Convert moles of unknown to grams (using molar mass).

Example: When 18.6 g of ethane (C2H6) burns, how many grams of CO2 are produced?
Equation: 2C2H6(g) + 7O2(g) → 4CO2(g) + 6H2O(g)
Calculation:
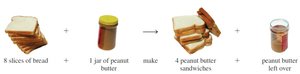
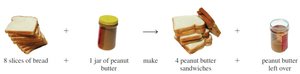
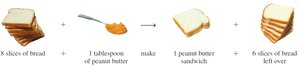
9.4 Limiting Reactants
In real chemical reactions, reactants are not always present in the exact ratios required by the balanced equation. The limiting reactant is the substance that is completely consumed first, thus limiting the amount of product formed.
Definition: The limiting reactant determines the maximum amount of product that can be formed.
Theoretical Yield: The amount of product formed when the limiting reactant is completely used up.
Percent Yield: A measure of the efficiency of a reaction, calculated as:
9.6 Energy in Chemical Reactions
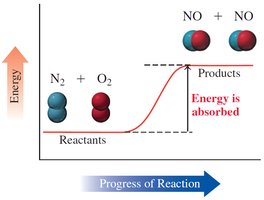
Chemical reactions involve energy changes, typically in the form of heat. The heat of reaction (ΔH) is the amount of heat absorbed or released at constant pressure.
Exothermic Reaction: Releases heat; ΔH is negative; products have less energy than reactants.
Endothermic Reaction: Absorbs heat; ΔH is positive; products have more energy than reactants.
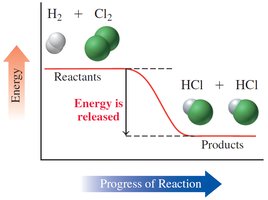

Example (Exothermic): ,
Example (Endothermic): ,
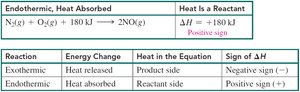
Summary Table:
Reaction | Energy Change | Heat in the Equation | Sign of ΔH |
|---|---|---|---|
Exothermic | Heat released | Product side | Negative (−) |
Endothermic | Heat absorbed | Reactant side | Positive (+) |
