 Back
BackLecture 13
Study Guide - Smart Notes

Chemical Reactions
Introduction to Chemical Reactions
Chemical reactions are fundamental processes in chemistry, involving the transformation of substances through the breaking and forming of chemical bonds. Understanding these reactions is essential for predicting product formation, energy changes, and the quantitative relationships between reactants and products.
Chemical reaction: A process in which one or more substances (reactants) are converted into one or more different substances (products).
Example: The reaction of minerals with dissolved CO2 to form carbonates, which plays a role in climate regulation.
Chemical Equations: The Language of Chemistry
Chemical equations are symbolic representations of chemical reactions, using formulas and symbols to describe the reactants and products involved. They provide a concise way to communicate chemical changes.
Reactants: Substances present before the reaction.
Products: Substances formed as a result of the reaction.
Arrow (→): Indicates the direction of the reaction, meaning "yields" or "produces".
State symbols: Indicate the physical state of each species: (s) = solid, (l) = liquid, (g) = gas, (aq) = aqueous.
Coefficients: Numbers placed in front of formulas to balance the equation; never change subscripts.
Example:
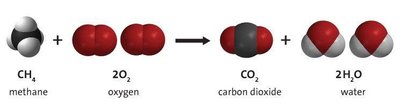
Balancing Chemical Equations
Balancing chemical equations ensures the conservation of mass, meaning the same number of atoms of each element are present on both sides of the equation. This is achieved by adjusting coefficients, not subscripts.
Balanced equation: Each element has the same number of atoms on both sides.
Steps: Identify reactants and products, count atoms, adjust coefficients as needed.
Example: Airbag reaction:

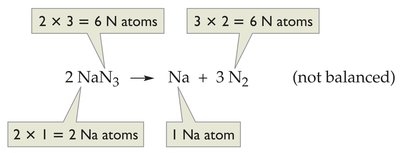
Stoichiometry: Quantitative Relationships in Reactions
Definition and Importance
Stoichiometry is the study of the quantitative relationships between reactants and products in a chemical reaction. It allows chemists to predict the amounts of substances consumed and produced.
Stoichiometry: From Greek for "element" and "measurement"; involves calculations based on balanced equations.
Coefficients: Represent the number of moles of each substance involved.
The Mole Concept
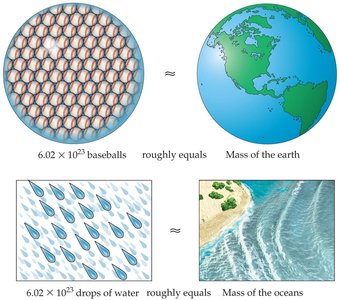
The mole is a fundamental unit in chemistry, used to count particles (atoms, molecules, ions) in a substance. It enables the conversion between mass, number of particles, and volume.
Mole (mol): The amount of substance containing particles (Avogadro's number).
Avogadro's number: , the number of atoms in 12 g of carbon-12.
Formula mass: The sum of atomic masses in a formula unit; for molecules, called molecular mass.
Volume Relationships in Chemical Equations
For reactions involving gases, the law of combining volumes and Avogadro's hypothesis are used to relate the volumes of reactants and products.
Law of combining volumes: At constant temperature and pressure, gaseous reactants and products combine in simple whole-number ratios.
Avogadro's hypothesis: Equal volumes of gases at the same temperature and pressure contain equal numbers of molecules.
Molar volume: One mole of any gas occupies 22.4 L at standard temperature and pressure (STP: 1 atm, 0°C).



Stoichiometric Calculations
Stoichiometric calculations involve converting between mass, moles, and volume using balanced equations and molar masses. The process follows a logical sequence of steps.
Step 1: Write and balance the chemical equation.
Step 2: Determine molar masses of substances involved.
Step 3: Use coefficients to convert moles of one substance to moles of another.
Step 4: Use molar mass to convert moles to grams.
Example: can be interpreted as "Two moles of hydrogen react with one mole of oxygen to produce two moles of water."

Summary Table: States of Matter in Chemical Equations
Symbol | State |
|---|---|
(s) | Solid |
(l) | Liquid |
(g) | Gas |
(aq) | Aqueous (dissolved in water) |
Key Takeaways
Chemical equations must be balanced to reflect the conservation of mass.
Stoichiometry allows quantitative predictions about reactants and products.
The mole is central to chemical calculations, connecting mass, volume, and number of particles.
Volume relationships in gas reactions are governed by simple ratios and Avogadro's hypothesis.
