 Back
BackLecture 14
Study Guide - Smart Notes

Chemical Reactions and Chemical Equations
Introduction to Chemical Reactions
Chemical reactions are processes in which substances (reactants) are transformed into new substances (products) through the breaking and forming of chemical bonds. Understanding chemical reactions is fundamental to chemistry, as it allows us to describe and predict changes in matter.
Reactants: The starting materials in a chemical reaction.
Products: The substances formed as a result of the reaction.
Chemical Equation: A symbolic representation of a chemical reaction, showing reactants and products with their respective quantities.
Balancing Chemical Equations
Balancing chemical equations ensures the law of conservation of mass is obeyed—meaning the number of atoms of each element is the same on both sides of the equation. This is essential for accurate chemical accounting and stoichiometric calculations.
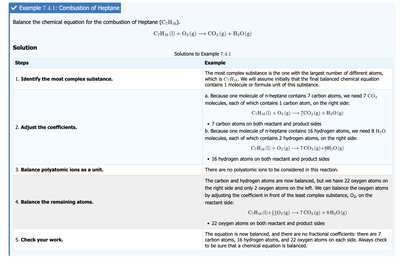
Step 1: Identify reactants and products and write the unbalanced equation.
Step 2: Make a table to count the number of atoms of each element on both sides.
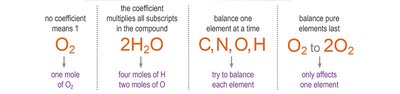
Step 3: Adjust coefficients (never subscripts) to balance one element at a time.
Step 4: Balance pure elements last and check your work.
Alternative Strategy: Start with the most complex substance, balance polyatomic ions as units, and use fractional coefficients if necessary, then multiply to clear fractions.
Example: Balancing the Photosynthesis Equation
Photosynthesis is a key environmental process that can be described by a chemical equation:
Unbalanced: CO2 + H2O → C6H12O6 + O2
Balanced: 6CO2 + 6H2O → C6H12O6 + 6O2
Molecular, Molar, and Mass Relationships in Chemical Equations
Understanding Stoichiometry
Stoichiometry is the quantitative study of reactants and products in a chemical reaction. It allows chemists to predict the amounts of substances consumed and produced in a reaction using balanced chemical equations.
Molecular Relationships: The coefficients in a balanced equation indicate the ratio of molecules involved.
Molar Relationships: The coefficients also represent the ratio of moles of each substance.
Mass Relationships: By using molar masses, we can convert between mass and moles for each substance.
Example: For the reaction 2NO + O2 → 2NO2:
2 molecules of NO react with 1 molecule of O2 to form 2 molecules of NO2.
2 moles of NO react with 1 mole of O2 to form 2 moles of NO2.

Steps in a Stoichiometric Calculation
Write and balance the chemical equation.
Determine the molar masses of all substances involved.
Use the coefficients to convert moles of the given substance to moles of the desired substance.
Convert moles to grams using molar mass.

Example: Molar Relationships
When 0.105 mol of propane (C3H8) is burned in excess oxygen, how many moles of O2 are consumed?
Balanced equation: C3H8 + 5 O2 → 3 CO2 + 4 H2O
From the equation, 1 mol C3H8 reacts with 5 mol O2.
So, 0.105 mol C3H8 × (5 mol O2 / 1 mol C3H8) = 0.525 mol O2
Example: Mass Relationships
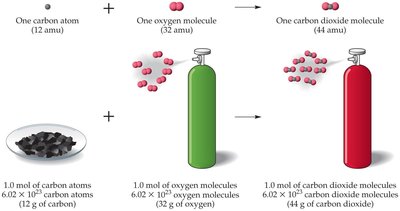
Calculate the mass of O2 needed to react with 5.00 g of carbon to form CO2:
Step 1: Convert grams of C to moles of C.
Step 2: Use the balanced equation to relate moles of C to moles of O2.
Step 3: Convert moles of O2 to grams of O2.
Calculation:
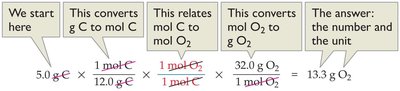
Solution Chemistry
What is a Solution?
A solution is a homogeneous mixture composed of two or more substances. The major component is called the solvent, and the minor component(s) are called solute(s).
Solvent: The substance present in the largest amount; it dissolves the solute.
Solute: The substance being dissolved.
Water is often referred to as the "universal solvent" due to its ability to dissolve many substances.

Solution Concentration
The concentration of a solution describes the amount of solute present in a given quantity of solvent or solution. Common ways to express concentration include molarity and percent concentration.
Dilute Solution: Contains a small amount of solute relative to solvent.
Concentrated Solution: Contains a large amount of solute relative to solvent.
Molarity (M)
Molarity is defined as the number of moles of solute per liter of solution:
Example: To prepare 0.750 L of 0.15 M NaCl solution, calculate the mass of NaCl required:
Moles NaCl = 0.15 mol/L × 0.750 L = 0.1125 mol
Mass NaCl = 0.1125 mol × 58.44 g/mol = 6.57 g

Percent Concentration
Percent concentration can be expressed by volume or by mass:
Percent by Volume:
Percent by Mass:
Example (Percent by Volume): What is the percent alcohol by volume (ABV) of a pint of beer (500 mL) that contains 25 mL alcohol?
Percent ABV = (25 mL / 500 mL) × 100 = 5%
Example (Percent by Mass): What is the percent sugar by mass of hummingbird food that contains 20 g sugar and 80 g water?
Total mass = 20 g + 80 g = 100 g
Percent by mass = (20 g / 100 g) × 100 = 20%
Solution Preparation Example: To make 430 g of an aqueous solution that is 4.85% by mass NaNO3:
Mass NaNO3 = 4.85% × 430 g = 20.86 g
Mass water = 430 g - 20.86 g = 409.14 g
Key Takeaways
Chemical accounting through balanced equations is essential for understanding reactions.
Stoichiometry connects molecular, molar, and mass relationships in chemical reactions.
Solution chemistry involves understanding the composition and concentration of mixtures.
