 Back
BackDynamic Equilibrium and Le Chatelier's Principle: Study Notes for Introductory Chemistry
Study Guide - Smart Notes

Dynamic Equilibrium
Closed System Reactions
Dynamic equilibrium occurs in reversible reactions within a closed system, where reactants and products are not lost to the surroundings. Understanding the nature of chemical systems is essential for predicting equilibrium behavior.
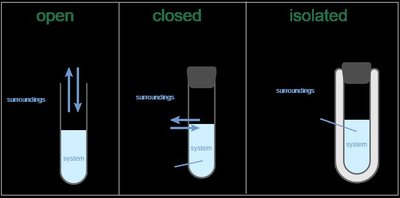
Open System: Both matter and energy can be exchanged with the surroundings.
Closed System: Only energy can be exchanged; matter remains within the system.
Isolated System: Neither matter nor energy is exchanged with the surroundings.
Relevance: Equilibrium can only be achieved in a closed system.
Reversible Reactions and Equilibrium Notation
Reversible reactions are those in which products can reform reactants. These reactions are represented by a double arrow (⇋), indicating that the process occurs in both directions and does not go to completion.
Notation: The double arrow (⇋) is used instead of a single arrow (→).
Example:
Reaching Equilibrium
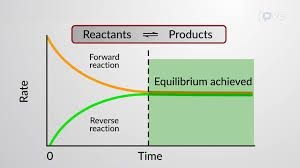
At the start of a reversible reaction, the forward reaction is fast due to a high concentration of reactants. As products accumulate, the reverse reaction rate increases. Eventually, both reactions occur at the same rate, and equilibrium is achieved.
Collision Theory: High concentration of reactants leads to frequent collisions and a fast forward reaction.
Equilibrium: The rates of the forward and reverse reactions become equal.
Mixture: At equilibrium, both reactants and products are present.
Equilibrium Position
The equilibrium position refers to the relative concentrations of reactants and products at equilibrium. These concentrations remain constant but are not necessarily equal.
Constant Concentrations: At equilibrium, concentrations do not change over time.
Not Equal: The concentrations of reactants and products are not always the same.

Dynamic Equilibrium
Dynamic equilibrium is a state where the forward and reverse reactions continue to occur at equal rates, so the concentrations of reactants and products remain constant. The system is dynamic because reactions are ongoing.
Constant Activity: Reactants are converted to products and vice versa simultaneously.
Independence: Equilibrium can be reached regardless of starting with reactants or products.

Controlling Equilibrium: Le Chatelier's Principle
Le Chatelier's Principle
Le Chatelier's Principle states that if a system at equilibrium is subjected to a change in conditions, the system will adjust to counteract that change and restore equilibrium.
Changes: Concentration, pressure, and temperature can affect equilibrium position.
Yield: Adjusting conditions can increase product yield and make processes more profitable.
Changing Concentration
Altering the concentration of reactants or products shifts the equilibrium position to counteract the change.
Add Reactant: Equilibrium shifts right (toward products).
Remove Reactant: Equilibrium shifts left (toward reactants).
Add Product: Equilibrium shifts left (toward reactants).
Remove Product: Equilibrium shifts right (toward products).
Changing Pressure (for Gaseous Reactions)
Pressure changes only affect reactions involving gases. Increasing pressure shifts equilibrium toward the side with fewer gas molecules, reducing pressure.
Increase Pressure: Shifts equilibrium to the side with fewer gas molecules.
Decrease Pressure: Shifts equilibrium to the side with more gas molecules.
Changing Temperature
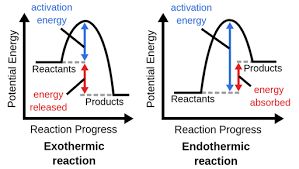
Temperature changes affect the direction of equilibrium depending on whether the reaction is exothermic or endothermic.
Exothermic Reaction: Releases heat in the forward direction; reverse is endothermic.
Increase Temperature: Shifts equilibrium in the endothermic direction (absorbs heat).
Decrease Temperature: Shifts equilibrium in the exothermic direction (releases heat).
The Role of Catalysts
Catalysts speed up the attainment of equilibrium but do not affect the position of equilibrium. They allow equilibrium to be reached faster and at lower temperatures, improving process efficiency.
Effect: No change in equilibrium position.
Benefit: Faster reaction rates and lower operational temperatures.
Equilibrium Constant
Expression for Equilibrium Constant (K)
The equilibrium constant (K) quantifies the ratio of product and reactant concentrations at equilibrium for a given reaction.
General Formula: For the reaction :

Summary Table: Effects on Equilibrium Position
Change | Equilibrium Shift | Effect |
|---|---|---|
Add Reactant | Right | More Product |
Remove Reactant | Left | More Reactant |
Add Product | Left | More Reactant |
Remove Product | Right | More Product |
Increase Pressure (gases) | Side with fewer gas molecules | Reduces pressure |
Increase Temperature | Endothermic direction | Absorbs heat |
Decrease Temperature | Exothermic direction | Releases heat |
Add Catalyst | No shift | Equilibrium reached faster |
Example: In the reaction , adding more hydrogen gas shifts equilibrium to the right, producing more water.
Additional info: Academic context was added to clarify the effects of changes in concentration, pressure, and temperature, and to provide a summary table for exam preparation.
