 Back
BackElectronic Structure and Periodic Properties of Elements
Study Guide - Smart Notes

Electromagnetic Energy and the Nature of Light
Wave Properties of Light
Light exhibits both wave-like and particle-like properties, a concept known as wave-particle duality. Understanding the behavior of light as a wave is fundamental to the study of atomic structure.
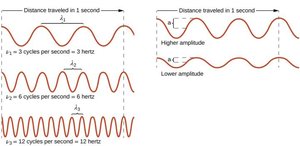
Wavelength (λ): The distance between two consecutive peaks or troughs in a wave.
Frequency (ν): The number of wave cycles that pass a given point per second, measured in hertz (Hz).
Amplitude: The height of the wave from its center to its peak (or trough), related to the wave's energy.
Speed of Light (c): In a vacuum, all electromagnetic waves travel at m/s.
Relationship:
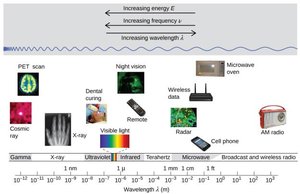
The Electromagnetic Spectrum
The electromagnetic spectrum encompasses all types of electromagnetic radiation, from gamma rays to radio waves. Visible light is only a small portion of this spectrum, with each color corresponding to a specific wavelength and frequency.
As wavelength increases, frequency and energy decrease.
Gamma rays have the shortest wavelength and highest energy; radio waves have the longest wavelength and lowest energy.
Interference and Standing Waves
Light waves can interfere with each other, producing patterns of constructive and destructive interference. This phenomenon is evidence of the wave nature of light and cannot be explained by classical particle theory.
Constructive interference: Occurs when wave peaks coincide, resulting in increased amplitude.
Destructive interference: Occurs when a peak coincides with a trough, canceling each other out.

Blackbody Radiation and Quantum Theory
Blackbody Radiation and the Ultraviolet Catastrophe
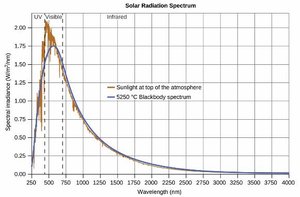
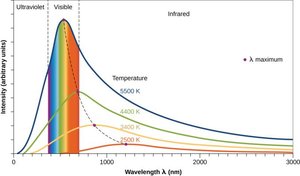
A blackbody is an idealized object that absorbs and emits all frequencies of electromagnetic radiation. Classical physics could not explain the observed spectrum of blackbody radiation, especially at short wavelengths (the ultraviolet catastrophe).
Experimental data showed that the intensity of radiation peaks at a certain wavelength and then decreases at shorter wavelengths.
Planck's Quantum Hypothesis
Max Planck resolved the ultraviolet catastrophe by proposing that energy is quantized and can only be emitted or absorbed in discrete amounts called quanta.
Planck's equation: where is an integer, is Planck's constant ( J·s), and is frequency.
The Bohr Model of the Atom
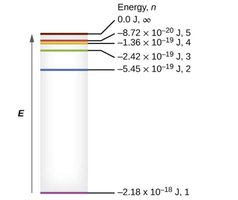
Energy Levels in the Hydrogen Atom
Niels Bohr proposed that electrons in a hydrogen atom occupy only certain allowed orbits with quantized energies. Electrons can move between these orbits by absorbing or emitting photons of specific energies.
Ground state: The lowest energy level (n = 1).
Excited state: Any energy level above the ground state (n > 1).
Energy of an orbit: , where is a constant.
Energy difference:
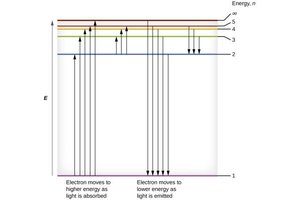
Electron Transitions and Spectral Lines
When an electron transitions between energy levels, it absorbs or emits a photon with energy equal to the difference between the two levels. This explains the line spectra observed for hydrogen and other elements.
Absorption: Electron moves to a higher energy level (n increases).
Emission: Electron falls to a lower energy level (n decreases).
Quantum Mechanical Model of the Atom
Quantum Numbers and Atomic Orbitals
The quantum mechanical model describes electrons as occupying orbitals, which are regions of space with a high probability of finding an electron. Each electron in an atom is described by four quantum numbers:
Principal quantum number (n): Indicates the energy level and size of the orbital (n = 1, 2, 3, ...).
Angular momentum quantum number (l): Defines the shape of the orbital (l = 0, 1, ..., n-1; s, p, d, f).
Magnetic quantum number (ml): Specifies the orientation of the orbital (ml = -l to +l).
Spin quantum number (ms): Describes the spin of the electron (ms = +½ or -½).
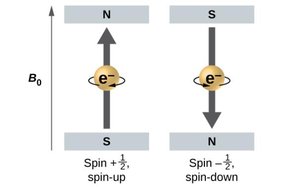
Pauli Exclusion Principle
No two electrons in the same atom can have the same set of four quantum numbers. This limits the number of electrons in each orbital to two, with opposite spins.
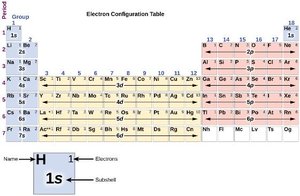
Electron Configurations and the Periodic Table
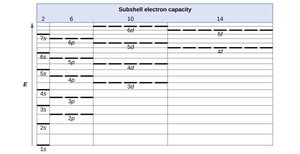
Orbital Energies and Electron Filling Order
Electrons fill atomic orbitals in order of increasing energy, following the Aufbau principle. The order is generally: 1s < 2s < 2p < 3s < 3p < 4s < 3d < 4p, etc. The Pauli exclusion principle and Hund's rule also govern electron arrangements.
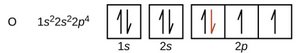
Electron Configuration Notation
The electron configuration of an atom is written using the principal quantum number, the subshell letter, and a superscript indicating the number of electrons in that subshell (e.g., 1s2 2s2 2p6).
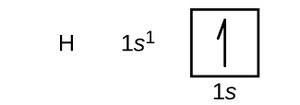
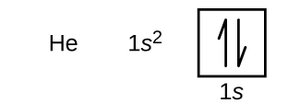
Orbital Diagrams
Orbital diagrams use boxes to represent orbitals and arrows to represent electrons. Upward arrows indicate spin +½, downward arrows indicate spin -½.
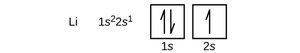
Core and Valence Electrons
Valence electrons are those in the outermost shell and are responsible for chemical reactivity. Core electrons are those in inner shells. Electron configurations can be abbreviated using the noble gas core notation.

Periodic Trends in Element Properties
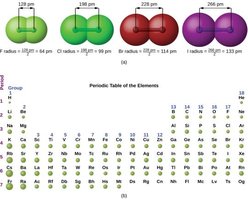
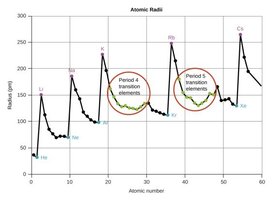
Atomic and Ionic Radii
The atomic radius increases down a group and decreases across a period due to changes in effective nuclear charge and principal quantum number. Ionic radius depends on whether the atom forms a cation (smaller) or anion (larger).
Ionization Energy
Ionization energy is the energy required to remove an electron from a gaseous atom. It decreases down a group and increases across a period, with some exceptions due to subshell structure and electron repulsion.
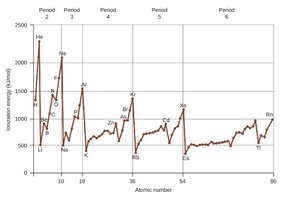
Exceptions in Ionization Energy Trends
Some elements deviate from the general trend due to electron configuration stability, such as the half-filled and fully filled subshells.
Electron Affinity
Electron affinity is the energy change when an atom gains an electron. It generally becomes more negative across a period, but there are exceptions for elements with filled or half-filled subshells.
