 Back
BackElectrons in Atoms and the Periodic Table: Structured Study Notes
Study Guide - Smart Notes

Electrons in Atoms and the Periodic Table
Atomic Models and Reactivity
The reactivity of elements, such as hydrogen, is explained by atomic models and their electron configurations. Group 1A elements (alkali metals) are highly reactive, while noble gases are inert due to their electron arrangements. The periodic law states that when elements are arranged by increasing atomic number, their properties recur periodically.
Bohr Model: Electrons travel in fixed orbits around the nucleus.
Quantum Mechanical Model: Electrons exist in orbitals defined by probability, not fixed paths.
Periodic Law: Properties of elements repeat in a predictable pattern.


Development of Atomic Models
Key scientists contributed to the development of quantum mechanics, which explains the behavior of electrons in atoms. These models help explain the periodic law and the chemical properties of elements.


Light and Electromagnetic Radiation
Nature of Light
Light is a form of electromagnetic radiation, not matter. It travels at a constant speed (c = 3.0 × 108 m/s) and exhibits both wave and particle properties.
Photon: A particle of light, carrying a specific amount of energy.
Electromagnetic Radiation: Energy that travels through space as waves.
Wave Properties of Light
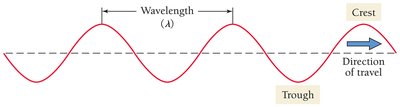
Light waves are characterized by wavelength (λ), frequency (ν), and amplitude. Wavelength is the distance between two consecutive crests.
Wavelength (λ): Measured in meters or nanometers.
Frequency (ν): Number of wave cycles per second (Hz).
Relationship: Wavelength and frequency are inversely related.

Visible Light and the Electromagnetic Spectrum
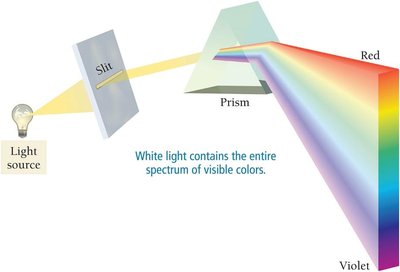
White light contains all visible wavelengths, which can be separated into colors (ROYGBIV) using a prism. Red light has the longest wavelength and lowest energy; violet has the shortest wavelength and highest energy.
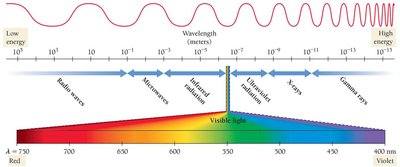
Visible Region: 400–750 nm.
Electromagnetic Spectrum: Includes radio waves, microwaves, infrared, visible, ultraviolet, X-rays, and gamma rays.


Energy and Frequency of Light
The energy of a photon depends on its wavelength and frequency. Shorter wavelengths (higher frequency) carry more energy.
Energy Formula: or
Planck's Constant (h): J·s
Speed of Light (c): m/s
Emission Spectra and Atomic Structure
Emission Spectra of Elements
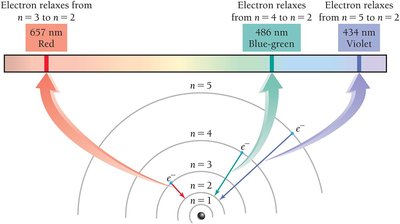
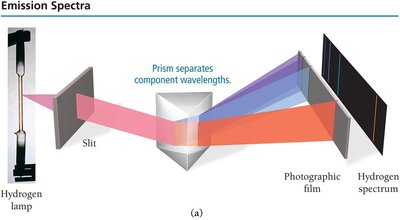
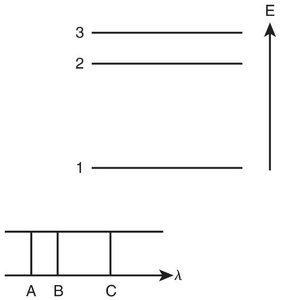
When atoms are energized, they emit light at characteristic wavelengths. Each element has a unique emission spectrum.
Continuous Spectrum: White light emits all wavelengths.
Line Spectrum: Elements emit only specific wavelengths.



The Bohr Model of the Atom
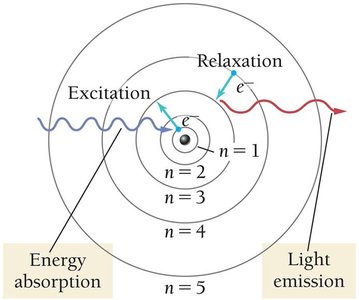
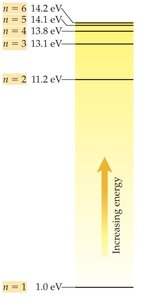
The Bohr model explains emission spectra by proposing quantized energy levels for electrons. Electrons can only occupy specific orbits (energy levels) and emit photons when transitioning between levels.
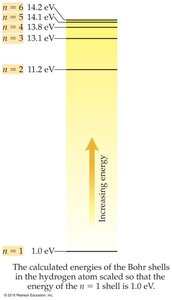
Quantum Number (n): Specifies the energy level.
Quantized Energy: Electrons cannot exist between orbits.

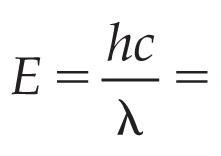
Energy Calculations
To calculate the energy of electromagnetic radiation:
Energy states:
Ground State: Lowest energy state, electrons closest to nucleus.
Excited State: Higher energy state, electrons farther from nucleus.
Quantum Mechanical Model of the Atom
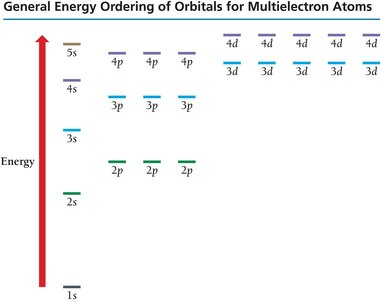
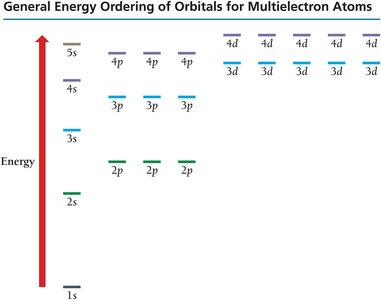
Quantum Numbers and Orbitals
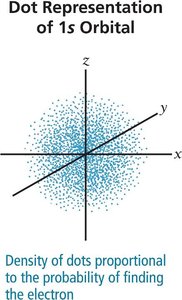
The quantum-mechanical model describes electrons in terms of orbitals, defined by quantum numbers:
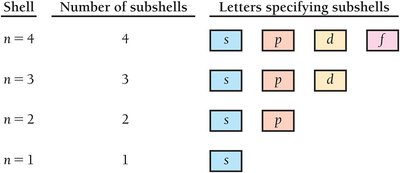
Principal Quantum Number (n): Specifies energy level (shell).
Subshells: Indicated by letters (s, p, d, f), each with a distinct shape.
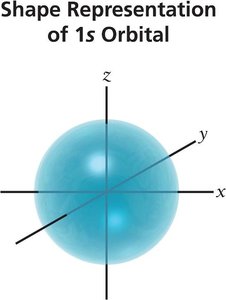
Orbital Shapes
Each subshell has a characteristic shape:
s orbital: Spherical
p orbital: Dumbbell-shaped
d orbital: Complex, four-lobed shapes
f orbital: Even more complex shapes
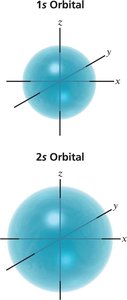
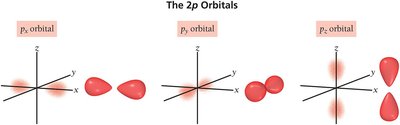
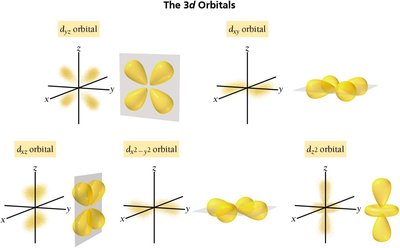
Electron Configurations
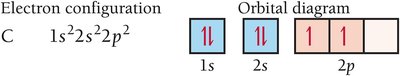
Writing Electron Configurations
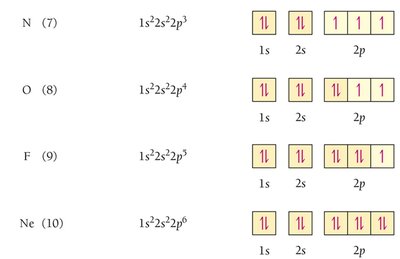
Electron configuration shows how electrons occupy orbitals. The ground-state configuration for hydrogen is 1s1.
Orbital Diagram: Uses arrows to represent electron spins.
Pauli Exclusion Principle: Orbitals hold no more than two electrons with opposite spins.
Hund’s Rule: Electrons fill orbitals singly before pairing.
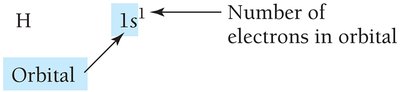
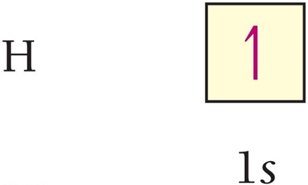



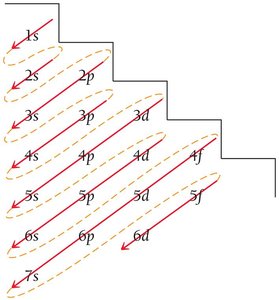
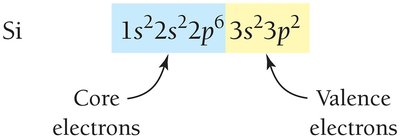
Noble Gas Core Notation
Electron configurations for elements beyond neon can be abbreviated using the noble gas core notation.
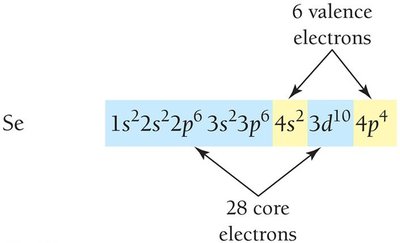
Valence and Core Electrons
Valence electrons are in the outermost shell and are involved in chemical bonding. Core electrons are in inner shells.
Periodic Table and Electron Configurations
Periodic Table Patterns
The periodic table is organized by electron configurations:
s block: First two groups on the left
p block: Six groups on the right
d block: Transition metals
f block: Lanthanides and actinides
Main-Group and Transition Elements
Main-group elements have valence electrons equal to their group number. Transition elements fill d orbitals, with exceptions for chromium and copper due to stability of half-filled and fully-filled subshells.
Periodic Trends
Atomic Size
Atomic size decreases across a period (left to right) due to increased nuclear charge, and increases down a group due to higher principal quantum number.
Ionization Energy
Ionization energy is the energy required to remove an electron. It increases across a period and decreases down a group.
Metallic Character
Metallic character increases down a group and decreases across a period. Metals tend to lose electrons and form cations.
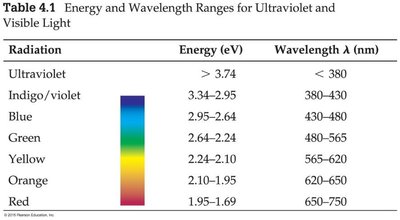
Summary Table: Energy and Wavelength Ranges for Visible Light
Radiation | Energy (eV) | Wavelength λ (nm) |
|---|---|---|
Ultraviolet | > 3.74 | < 380 |
Indigo/violet | 3.34–2.95 | 380–430 |
Blue | 2.95–2.64 | 430–480 |
Green | 2.64–2.24 | 480–565 |
Yellow | 2.24–2.10 | 565–620 |
Orange | 2.10–1.95 | 620–650 |
Red | 1.95–1.69 | 650–750 |

Additional info: Academic context and explanations have been expanded for clarity and completeness.
