 Back
BackElectrons in Atoms and the Periodic Table: Models, Configurations, and Periodic Trends
Study Guide - Smart Notes

Electrons in Atoms and the Periodic Table
Introduction to Atomic Models and Reactivity
The arrangement of electrons in atoms determines the chemical and physical properties of elements. Understanding why some elements are reactive (like hydrogen) and others are inert (like helium) requires models of atomic structure. These models also explain the periodic trends observed in the periodic table.


Hydrogen is highly reactive due to its single electron, which seeks stability through bonding.
Helium is inert because its two electrons fill its only shell, creating a stable configuration.
The periodic law states that properties of elements recur periodically when arranged by atomic number.
Models of the Atom
The Bohr Model
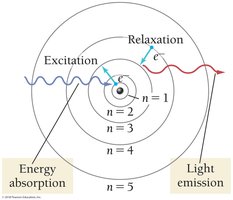
The Bohr model describes electrons as moving in fixed orbits around the nucleus, each with a specific energy. This model successfully explains the emission spectrum of hydrogen but fails for more complex atoms.
Each orbit is associated with a quantum number n (n = 1, 2, 3, ...).
Electrons can move between orbits by absorbing or emitting energy as photons.

The Quantum-Mechanical Model
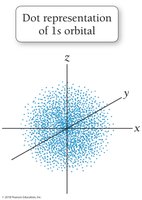
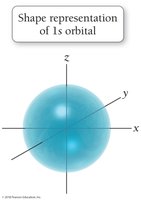
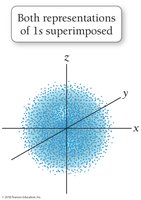
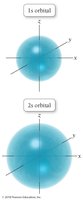
The quantum-mechanical model replaces orbits with orbitals, which are probability maps describing where electrons are likely to be found. This model accounts for the behavior of all elements and explains periodic trends.
Orbitals are defined by quantum numbers and have specific shapes (s, p, d, f).
Electrons do not have exact paths but exist in regions of high probability.
Light and Electromagnetic Radiation
Nature of Light
Light is a form of electromagnetic radiation, exhibiting both wave-like and particle-like properties. The interaction of light with atoms is fundamental to understanding atomic structure.
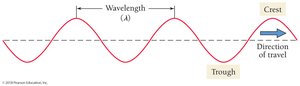
Wavelength (λ): Distance between adjacent wave crests.
Frequency (ν): Number of wave cycles passing a point per second.
Wavelength and frequency are inversely related: where is the speed of light.
Light energy is quantized in packets called photons:
The Electromagnetic Spectrum
The electromagnetic spectrum includes all types of electromagnetic radiation, from gamma rays (shortest wavelength, highest energy) to radio waves (longest wavelength, lowest energy). Visible light is only a small part of the spectrum.
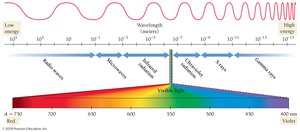
Gamma rays and X-rays have enough energy to damage biological molecules.
Ultraviolet (UV) light can cause sunburn and increase cancer risk.
Visible light enables vision but does not damage molecules.
Infrared, microwaves, and radio waves have progressively lower energy.

Color and Atomic Emission Spectra
Each element emits light at specific wavelengths, producing a unique atomic emission spectrum. This property is used in identifying elements and understanding atomic structure.

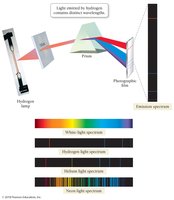
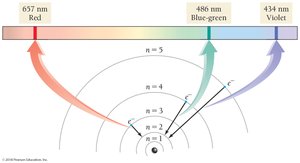
Electron Transitions and Spectra
Quantized Energy Levels
Electrons in atoms occupy quantized energy levels. When an electron transitions between levels, it absorbs or emits a photon with energy equal to the difference between the levels.
Energy difference:
Photon energy:
Quantum Numbers and Orbitals
Principal Quantum Number (n) and Subshells
The principal quantum number (n) determines the energy and size of an orbital. Each shell contains subshells (s, p, d, f) with characteristic shapes and energies.

s orbitals: Spherical shape
p orbitals: Dumbbell shape
d and f orbitals: More complex shapes
Electron Configurations
Writing Electron Configurations
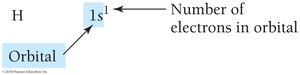
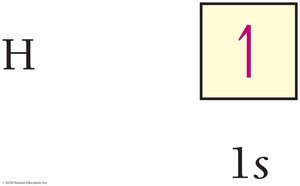
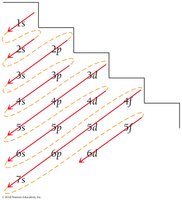
Electron configurations describe the arrangement of electrons in an atom's orbitals. The order of filling is determined by increasing energy, following the Aufbau principle, Pauli exclusion principle, and Hund's rule.
Aufbau principle: Lower-energy orbitals fill before higher-energy orbitals.
Pauli exclusion principle: Each orbital holds a maximum of two electrons with opposite spins.
Hund's rule: Electrons occupy orbitals singly before pairing.




Noble Gas Notation and Valence Electrons
Electron configurations for larger atoms can be abbreviated using the noble gas core notation. Valence electrons are those in the outermost shell and are responsible for chemical reactivity.

Core electrons: Inner shells, not involved in bonding
Valence electrons: Outermost shell, involved in bonding
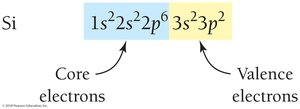
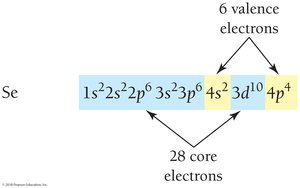
Electron Configurations and the Periodic Table
Blocks and Patterns
The periodic table is divided into blocks (s, p, d, f) corresponding to the type of orbital being filled. Elements in the same group have similar valence electron configurations and chemical properties.
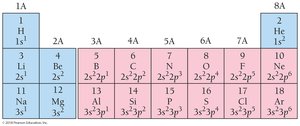

Periodic Trends in Electron Configurations
Main-group elements: Number of valence electrons equals the group number.
Transition metals: d orbitals are filled; exceptions exist (e.g., Cr, Cu).
Periodic Trends
Atomic Size
Atomic size decreases across a period (left to right) due to increasing nuclear charge and increases down a group due to higher principal quantum numbers.
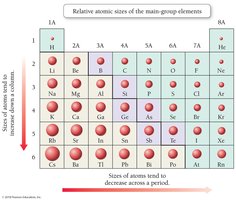
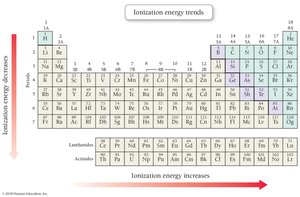
Ionization Energy
Ionization energy is the energy required to remove an electron from an atom. It increases across a period and decreases down a group.
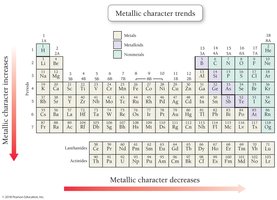
Metallic Character
Metallic character refers to the tendency to lose electrons. It decreases across a period and increases down a group.
Special Groups and Reactivity
Noble Gases
Noble gases have full valence shells, making them chemically inert. Their stability explains the reactivity of other elements seeking to achieve similar configurations.

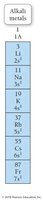
Alkali Metals
Alkali metals (Group 1) have one valence electron and are highly reactive, tending to lose one electron to form +1 cations.
Alkaline Earth Metals
Alkaline earth metals (Group 2) have two valence electrons and tend to lose both to form +2 cations.

Halogens
Halogens (Group 7) have seven valence electrons and tend to gain one electron to form -1 anions, achieving noble gas configurations.

Summary Table: Electron Configuration and Periodic Trends
Trend | Across a Period (→) | Down a Group (↓) |
|---|---|---|
Atomic Size | Decreases | Increases |
Ionization Energy | Increases | Decreases |
Metallic Character | Decreases | Increases |
Key Learning Objectives
Understand electromagnetic radiation and its interaction with atoms.
Describe the Bohr and quantum-mechanical models of the atom.
Write electron configurations and orbital diagrams for elements.
Identify valence and core electrons.
Explain periodic trends in atomic size, ionization energy, and metallic character.
